
天津医药 ›› 2023, Vol. 51 ›› Issue (6): 573-579.doi: 10.11958/20221405
收稿日期:2022-09-02
修回日期:2022-10-21
出版日期:2023-06-15
发布日期:2023-06-20
通讯作者:
△E-mail:作者简介:杜文峰(1987),男,主治医师,主要从事肺癌、慢性阻塞性肺疾病及哮喘方面研究。E-mail:基金资助:
DU Wenfeng( ), ZHOU Ling, LI Qi(
), ZHOU Ling, LI Qi( )
)
Received:2022-09-02
Revised:2022-10-21
Published:2023-06-15
Online:2023-06-20
Contact:
△E-mail:杜文峰, 周玲, 李琪. CircCDR1as调节miR-671-5p/CBX4轴对NSCLC细胞生物学行为的影响[J]. 天津医药, 2023, 51(6): 573-579.
DU Wenfeng, ZHOU Ling, LI Qi. Effects of CircCDR1as on the biological behavior of NSCLC cells by regulating the miR-671-5p/CBX4 axis[J]. Tianjin Medical Journal, 2023, 51(6): 573-579.
摘要:
目的 探讨环状RNA(CircRNA)反义小脑变性相关蛋白1转录本(CircCDR1as)通过调节miR-671-5p/染色盒同源物4(CBX4)轴对非小细胞肺癌(NSCLC)细胞增殖、凋亡、迁移和侵袭的影响。方法 体外培养人NSCLC细胞株(NCI-H524、NCI-H1734、Calu-3和A549)和人支气管上皮样细胞(HBE)。A549细胞分成Control组、si-NC组、si-CircCDR1as组、si-CircCDR1as+anti-miR-NC组、si-CircCDR1as+anti-miR-671-5p组、pcDNA组、CircCDR1as组、miR-NC组、miR-671-5p组、miR-671-5p+pcDNA组、miR-671-5p+CBX4组、anti-miR-NC组和anti-miR-671-5p组。采用实时定量聚合酶链反应(qRT-PCR)检测CircCDR1as、miR-671-5p和CBX4 mRNA表达。Western blot检测CBX4蛋白表达。分别采用四甲基偶氮唑盐(MTT)、克隆形成实验、流式细胞术和Transwell检测细胞活力、增殖、凋亡、迁移和侵袭。通过双荧光素酶报告基因检测验证miR-671-5p与CircCDR1as或CBX4之间的靶向关系。结果 与HBE细胞相比,CircCDR1as和CBX4在4种NSCLC细胞中表达升高,miR-671-5p表达降低(P<0.05)。与si-NC组比较,si-CircCDR1as组A549细胞活力和克隆形成数显著降低,细胞迁移和侵袭数目减少,凋亡率增高(P<0.05)。miR-671-5p作为CircCDR1as的靶点,其下调减弱了CircCDR1as沉默对NSCLC进展的调控作用(P<0.05)。miR-671-5p靶向CBX4在体外抑制NSCLC的恶性进展(P<0.05)。沉默CircCDR1as可通过上调miR-671-5p水平降低CBX4的表达(P<0.05)。结论 CircCDR1as沉默可通过上调miR-671-5p和下调CBX4表达来抑制细胞增殖、迁移和侵袭,诱导细胞凋亡。
中图分类号:
| 细胞系 | CircCDR1as | miR-671-5p | CBX4 mRNA | CBX4蛋白 |
|---|---|---|---|---|
| HBE | 1.00±0.01 | 1.00±0.02 | 1.00±0.02 | 1.00±0.03 |
| NCI-H524 | 3.92±0.14a | 0.59±0.05a | 2.86±0.14a | 2.54±0.11a |
| NCI-H1734 | 4.09±0.21a | 0.52±0.06a | 2.95±0.18a | 2.63±0.14a |
| Calu-3 | 4.15±0.19a | 0.41±0.04a | 3.18±0.23a | 2.75±0.21a |
| A549 | 4.29±0.28a | 0.32±0.02a | 3.32±0.25a | 2.89±0.16a |
| F | 328.965** | 243.424** | 160.266** | 175.065** |
表1 HBE细胞和NSCLC细胞系中CircCDR1as、miR-671-5p、CBX4表达水平 (n=6,$\bar{x}±s$)
Tab.1 CircCDR1as, miR-671-5p and CBX4 levels in HBE cells and NSCLC cell lines
| 细胞系 | CircCDR1as | miR-671-5p | CBX4 mRNA | CBX4蛋白 |
|---|---|---|---|---|
| HBE | 1.00±0.01 | 1.00±0.02 | 1.00±0.02 | 1.00±0.03 |
| NCI-H524 | 3.92±0.14a | 0.59±0.05a | 2.86±0.14a | 2.54±0.11a |
| NCI-H1734 | 4.09±0.21a | 0.52±0.06a | 2.95±0.18a | 2.63±0.14a |
| Calu-3 | 4.15±0.19a | 0.41±0.04a | 3.18±0.23a | 2.75±0.21a |
| A549 | 4.29±0.28a | 0.32±0.02a | 3.32±0.25a | 2.89±0.16a |
| F | 328.965** | 243.424** | 160.266** | 175.065** |
| 组别 | CircCDR1as | miR-671-5p | 细胞活力(OD490值) | 克隆形成数(个) | 凋亡率 (%) | 迁移细胞数(个) | 侵袭细胞数(个) |
|---|---|---|---|---|---|---|---|
| Control组 | 1.00±0.03 | 1.00±0.02 | 1.18±0.21 | 91.45±7.62 | 10.02±0.91 | 94.38±8.12 | 97.56±6.89 |
| si-NC组 | 1.00±0.02 | 0.99±0.03 | 1.17±0.19 | 91.38±7.49 | 10.01±0.89 | 94.29±7.91 | 97.54±6.85 |
| si-CircCDR1as组 | 0.38±0.03ab | 1.78±0.06ab | 0.49±0.07ab | 38.62±3.51ab | 33.45±2.62ab | 40.15±10.03ab | 41.32±8.74ab |
| si-CircCDR1as+anti-miR-NC组 | 0.39±0.02 | 1.75±0.08 | 0.47±0.06 | 38.67±3.48 | 33.48±2.14 | 40.18±10.06 | 41.35±6.95 |
| si-CircCDR1as+anti-miR-671-5p组 | 0.91±0.05cd | 1.26±0.05cd | 0.99±0.12cd | 80.59±6.72cd | 14.13±1.06cd | 79.84±9.21cd | 80.69±7.24cd |
| F | 611.941** | 328.761** | 36.693** | 121.564** | 315.182** | 55.305** | 90.091** |
表2 各组NSCLC细胞活力、克隆形成数、凋亡率、迁移侵袭细胞数比较 (n=6,$\bar{x}±s$)
Tab.2 Comparison of NSCLC cell viability, number of clone formation, apoptosis rate, migration and number of invasive cells between the five groups
| 组别 | CircCDR1as | miR-671-5p | 细胞活力(OD490值) | 克隆形成数(个) | 凋亡率 (%) | 迁移细胞数(个) | 侵袭细胞数(个) |
|---|---|---|---|---|---|---|---|
| Control组 | 1.00±0.03 | 1.00±0.02 | 1.18±0.21 | 91.45±7.62 | 10.02±0.91 | 94.38±8.12 | 97.56±6.89 |
| si-NC组 | 1.00±0.02 | 0.99±0.03 | 1.17±0.19 | 91.38±7.49 | 10.01±0.89 | 94.29±7.91 | 97.54±6.85 |
| si-CircCDR1as组 | 0.38±0.03ab | 1.78±0.06ab | 0.49±0.07ab | 38.62±3.51ab | 33.45±2.62ab | 40.15±10.03ab | 41.32±8.74ab |
| si-CircCDR1as+anti-miR-NC组 | 0.39±0.02 | 1.75±0.08 | 0.47±0.06 | 38.67±3.48 | 33.48±2.14 | 40.18±10.06 | 41.35±6.95 |
| si-CircCDR1as+anti-miR-671-5p组 | 0.91±0.05cd | 1.26±0.05cd | 0.99±0.12cd | 80.59±6.72cd | 14.13±1.06cd | 79.84±9.21cd | 80.69±7.24cd |
| F | 611.941** | 328.761** | 36.693** | 121.564** | 315.182** | 55.305** | 90.091** |
| 组别 | CBX4 mRNA | CBX4蛋白 |
|---|---|---|
| miR-NC组 | 1.00±0.01 | 1.00±0.03 |
| miR-671-5p组 | 0.38±0.04a | 0.42±0.04a |
| anti-miR-NC组 | 1.00±0.02 | 1.00±0.02 |
| anti-miR-671-5p组 | 2.12±0.06b | 1.71±0.08b |
| F | 2 212.351** | 719.376** |
表3 各组A549细胞中CBX4的mRNA和蛋白水平比较 (n=6,$\bar{x}±s$)
Tab.3 Comparison of mRNA and protein levels of CBX4 in A549 cells between the four groups
| 组别 | CBX4 mRNA | CBX4蛋白 |
|---|---|---|
| miR-NC组 | 1.00±0.01 | 1.00±0.03 |
| miR-671-5p组 | 0.38±0.04a | 0.42±0.04a |
| anti-miR-NC组 | 1.00±0.02 | 1.00±0.02 |
| anti-miR-671-5p组 | 2.12±0.06b | 1.71±0.08b |
| F | 2 212.351** | 719.376** |
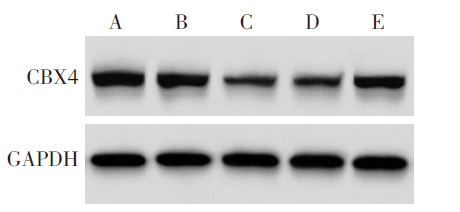
图6 各组A549细胞中CBX4蛋白表达 A:Control组;B:miR-NC组;C:miR-671-5p组;D:miR-671-5p+pcDNA组;E:miR-671-5p+CBX4组。
Fig.6 Expression of CBX4 protein in A549 cells of each group
| 组别 | CBX4 mRNA | CBX4蛋白 | 细胞活力(OD值) | 克隆细胞数 | 凋亡率(%) | 迁移细胞数 | 侵袭细胞数 |
|---|---|---|---|---|---|---|---|
| Control组 | 1.00±0.02 | 1.00±0.03 | 1.09±0.15 | 97.68±5.49 | 8.24±0.13 | 86.34±3.52 | 78.69±2.63 |
| miR-NC组 | 0.99±0.03 | 1.01±0.05 | 1.07±0.13 | 97.75±5.63 | 8.26±0.15 | 86.38±3.61 | 78.72±2.62 |
| miR-671-5p组 | 0.36±0.03ab | 0.33±0.02ab | 0.46±0.05ab | 39.54±2.16ab | 34.52±2.31ab | 40.15±2.03ab | 35.42±1.89ab |
| miR-671-5p+pcDNA组 | 0.37±0.02 | 0.32±0.04 | 0.48±0.06 | 39.57±2.13 | 34.39±2.46 | 40.19±2.05 | 35.51±1.92 |
| miR-671-5p+CBX4组 | 0.84±0.05cd | 0.81±0.05cd | 1.03±0.11cd | 82.36±4.25cd | 13.64±1.02cd | 81.23±3.12cd | 67.49±2.01cd |
| F | 613.941** | 455.051** | 55.276** | 297.408** | 441.727** | 412.551** | 585.024** |
表4 各组NSCLC细胞活力、克隆形成数、凋亡率、迁移和侵袭细胞数比较 (n=6,$\bar{x}±s$)
Tab.4 Comparison of NSCLC cell viability, number of clone formation, apoptosis rate, migration and number of invasive cells between the five groups
| 组别 | CBX4 mRNA | CBX4蛋白 | 细胞活力(OD值) | 克隆细胞数 | 凋亡率(%) | 迁移细胞数 | 侵袭细胞数 |
|---|---|---|---|---|---|---|---|
| Control组 | 1.00±0.02 | 1.00±0.03 | 1.09±0.15 | 97.68±5.49 | 8.24±0.13 | 86.34±3.52 | 78.69±2.63 |
| miR-NC组 | 0.99±0.03 | 1.01±0.05 | 1.07±0.13 | 97.75±5.63 | 8.26±0.15 | 86.38±3.61 | 78.72±2.62 |
| miR-671-5p组 | 0.36±0.03ab | 0.33±0.02ab | 0.46±0.05ab | 39.54±2.16ab | 34.52±2.31ab | 40.15±2.03ab | 35.42±1.89ab |
| miR-671-5p+pcDNA组 | 0.37±0.02 | 0.32±0.04 | 0.48±0.06 | 39.57±2.13 | 34.39±2.46 | 40.19±2.05 | 35.51±1.92 |
| miR-671-5p+CBX4组 | 0.84±0.05cd | 0.81±0.05cd | 1.03±0.11cd | 82.36±4.25cd | 13.64±1.02cd | 81.23±3.12cd | 67.49±2.01cd |
| F | 613.941** | 455.051** | 55.276** | 297.408** | 441.727** | 412.551** | 585.024** |
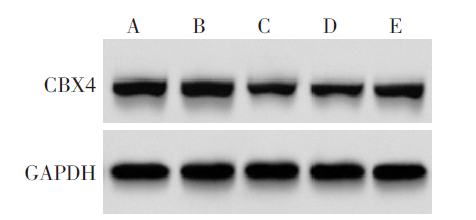
图8 各组A549细胞中CBX4蛋白表达 A:Control组;B:si-NC组;C:si-CircCDR1as组;D:si-CircCDR1as+anti-miR-NC组;E:si-CircCDR1as+anti-miR-671-5p组。
Fig.8 CBX4 protein expression in A549 cells of each group
| 组别 | CBX4 mRNA | CBX4蛋白 |
|---|---|---|
| Control组 | 1.00±0.03 | 1.00±0.02 |
| si-NC组 | 1.02±0.05 | 1.01±0.03 |
| si-CircCDR1as组 | 0.43±0.03ab | 0.49±0.05ab |
| si-CircCDR1as+anti-miR-NC组 | 0.41±0.02 | 0.50±0.07 |
| si-CircCDR1as+anti-miR-671-5p组 | 0.82±0.06cd | 0.87±0.09cd |
| F | 322.880** | 121.304** |
表5 各组NSCLC细胞CBX4表达的比较 (n=6,$\bar{x}±s$)
Tab.5 Comparison of CBX4 expression in NSCLC cells between the five groups
| 组别 | CBX4 mRNA | CBX4蛋白 |
|---|---|---|
| Control组 | 1.00±0.03 | 1.00±0.02 |
| si-NC组 | 1.02±0.05 | 1.01±0.03 |
| si-CircCDR1as组 | 0.43±0.03ab | 0.49±0.05ab |
| si-CircCDR1as+anti-miR-NC组 | 0.41±0.02 | 0.50±0.07 |
| si-CircCDR1as+anti-miR-671-5p组 | 0.82±0.06cd | 0.87±0.09cd |
| F | 322.880** | 121.304** |
| [1] | HIRSCH F R, SCAGLIOTTI G V, MULSHINE J L, et al. Lung cancer:current therapies and new targeted treatments[J]. Lancet, 2017, 389(10066):299-311. doi:10.1016/S0140-6736(16)30958-8. |
| [2] | HERBST R S, MORGENSZTERN D, BOSHOFF C. The biology and management of non-small cell lung cancer[J]. Nature, 2018, 553(7689):446-454. doi:10.1038/nature25183. |
| [3] | BALATA H, FONG K M, HENDRIKS L E, et al. Prevention and early detection for NSCLC:Advances in thoracic oncology 2018[J]. J Thorac Oncol, 2019, 14(9):1513-1527. doi:10.1016/j.jtho.2019.06.011. |
| [4] | TAN S, GOU Q, PU W, et al. Circular RNA F-circEA produced from EML4-ALK fusion gene as a novel liquid biopsy biomarker for non-small cell lung cancer[J]. Cell Res, 2018, 28(6):693-695. doi:10.1038/s41422-018-0033-7. |
| [5] | 安翠红, 宁文君, 马彩虹. 血清鳞状细胞癌相关抗原检测在超早期肺癌诊断中的应用价值[J]. 实用癌症杂志, 2019, 34(4):600-602. |
| AN C H, NING W J, MA C H. Diagnostic value of serum squamous cell carcinoma associated antigen in the diagnosis of ultra early lung cancer[J]. Journal of Practical Cancer, 2019, 34(4):600-602. doi:10.3969/j.issn.1001-5930.2019.04.023. | |
| [6] | BRAICU C, ZIMTA A A, HARANGUS A, et al. The function of non-coding RNAs in lung cancer tumorigenesis[J]. Cancers(Basel), 2019, 11(5):605-623. doi:10.3390/cancers11050605. |
| [7] | WEI H, LI L, ZHANG H, et al. Circ-FOXM1 knockdown suppresses non-small cell lung cancer development by regulating the miR-149-5p/ATG5 axis[J]. Cell Cycle, 2021, 20(2):166-178. doi:10.1080/15384101.2020.1867780. |
| [8] | NIU Y, HE J H, ZHANG Y, et al. Effect of the CircCDR1as/miR-641/XIAP regulatory axis on the proliferation and invasion of the prostate cancer PC-3 cell line[J]. Oncol Lett, 2021, 21(6):469-479. doi:10.3892/ol.2021.12730. |
| [9] | XIONG X, FENG J, YANG X, et al. Circular RNA CDR1as promotes tumor progression by regulating miR-432-5p/E2F3 axis in pancreatic cancer[J]. Cancer Cell Int, 2021, 21(1):112-122. doi:10.1186/s12935-021-01812-3. |
| [10] | LI P, YANG X, YUAN W, et al. CircRNA-Cdr1as exerts anti-oncogenic functions in bladder cancer by sponging microRNA-135a[J]. Cell Physiol Biochem, 2018, 46(4):1606-1616. doi:10.1159/000489208. |
| [11] | CHEN H, MAO M, JIANG J, et al. Circular RNA CDR1as acts as a sponge of miR-135b-5p to suppress ovarian cancer progression[J]. Onco Targets Ther, 2019, 12(1):3869-3879. doi:10.2147/OTT.S207938. |
| [12] | ZHANG X, YANG D, WEI Y. Overexpressed CDR1as functions as an oncogene to promote the tumor progression via miR-7 in non-small-cell lung cancer[J]. Onco Targets Ther, 2018, 11(1):3979-3987. doi:10.2147/OTT.S158316. |
| [13] | YE J, LUO W, LUO L, et al. MicroRNA-671-5p inhibits cell proliferation, migration and invasion in non-small cell lung cancer by targeting MFAP3L[J]. Mol Med Rep, 2022, 25(1):30-38. doi:10.3892/mmr.2021.12546. |
| [14] | ZHAO X, WU X. Polycomb-group proteins in the initiation and progression of cancer[J]. J Genet Genomics, 2021, 48(6):433-443. doi:10.1016/j.jgg.2021.03.013. |
| [15] | HU C, ZHANG Q, TANG Q, et al. CBX4 promotes the proliferation and metastasis via regulating BMI-1 in lung cancer[J]. J Cell Mol Med, 2020, 24(1):618-631. doi:10.1111/jcmm.14771. |
| [16] | 李莹, 孙万仆, 安志强, 等. 微小RNA-671-5p对人结肠癌细胞增殖、迁移和侵袭的影响[J]. 安徽医药, 2020, 24(5):942-946. |
| LI Y, SUN W P, AN Z Q, et al. miR-671-5p inhibits colon cancer cell proliferation,migration and invasion by targeting CBX4[J]. Anhui Med Pharm J, 2020, 24(5):942-946. doi:10.3969/j.issn.1009-6469.2020.05.024. | |
| [17] | ZHANG M, XIN Y. Circular RNAs:A new frontier for cancer diagnosis and therapy[J]. J Hematol Oncol, 2018, 11(1):21-30. doi:10.1186/s13045-018-0569-5. |
| [18] | ALTORKI N K, MARKOWITZ G J, GAO D, et al. The lung microenvironment: an important regulator of tumour growth and metastasis[J]. Nat Rev Cancer, 2019, 19(1):9-31. doi:10.1038/s41568-018-0081-9. |
| [19] | LI Y, ZHANG J, PAN S, et al. CircRNA CDR1as knockdown inhibits progression of non-small-cell lung cancer by regulating miR-219a-5p/SOX5 axis[J]. Thorac Cancer, 2020, 11(3):537-548. doi:10.1111/1759-7714.13274. |
| [20] | LEGRAS A, PÉCUCHET N, IMBEAUD S, et al. Epithelial-to-mesenchymal transition and microRNAs in lung cancer[J]. Cancers (Basel), 2017, 9(8):101-130. doi:10.3390/cancers9080101. |
| [21] | DONG Y, HE D, PENG Z, et al. Circular RNAs in cancer: an emerging key player[J]. J Hematol Oncol, 2017, 10(1):2-10. doi:10.1186/s13045-016-0370-2. |
| [22] | YANG W, YANG X, WANG X, et al. Silencing CDR1as enhances the sensitivity of breast cancer cells to drug resistance by acting as a miR-7 sponge to down-regulate REGγ[J]. J Cell Mol Med, 2019, 23(8):4921-4932. doi:10.1111/jcmm.14305. |
| [23] | GAO L, DOU Z C, REN W H, et al. CircCDR1as upregulates autophagy under hypoxia to promote tumor cell survival via AKT/ERK1/2/mTOR signaling pathways in oral squamous cell carcinomas[J]. Cell Death Dis, 2019, 10(10):745-761. doi:10.1038/s41419-019-1971-9. |
| [24] | 许英杰, 张艳炜, 李海洲, 等. miR-671-5p对非小细胞肺癌细胞增殖和迁移及侵袭影响及其作用机制[J]. 中华肿瘤防治杂志, 2021, 28(20):1541-1548. |
| XU Y J, ZHANG Y W, LI H Z, et al. Effects and mechanism of miR-671-5p on proliferation,migration and invasion of non-small cell lung cancer cells[J]. Chin J Cancer Prev Treat, 2021, 28(20):1541-1548. doi:10.16073/j.cnki.cjcpt.2021.20.05. | |
| [25] | MA C, NIE Z K, GUO H M, et al. miR-671-5p plays a promising role in restraining osteosarcoma cell characteristics through targeting TUFT1[J]. J Biochem Mol Toxicol, 2020, 34(7):e22490-e22498. doi:10.1002/jbt.22490. |
| [26] | ZENG J S, ZHANG Z D, PEI L, et al. CBX4 exhibits oncogenic activities in breast cancer via Notch1 signaling[J]. Int J Biochem Cell Biol, 2018, 95(1):1-8. doi:10.1016/j.biocel.2017.12.006. |
| [27] | WEN L J, WANG Y S, TAN P Y. miR-515-5p inhibits the proliferation,migration and invasion of human breast cancer cells by targeting CBX4[J]. Exp Ther Med, 2021, 22(5):1328-1337. doi:10.3892/etm.2021.10763. |
| [28] | PAN Y, LI Q, CAO Z, et al. The SUMO E3 ligase CBX4 is identified as a poor prognostic marker of gastric cancer through multipronged OMIC analyses[J]. Genes Dis, 2020, 8(6):827-837. doi:10.1016/j.gendis.2020.08.010. |
| [1] | 朱海燕, 王烨, 尹艳. 老年NSCLC患者术后胃肠功能紊乱的危险因素研究[J]. 天津医药, 2026, 54(3): 289-294. |
| [2] | 黄熷远, 付靖, 赵亚, 王龙灏, 仓顺东. 非小细胞肺癌EGFR-TKI耐药与p53基因突变的研究进展[J]. 天津医药, 2026, 54(3): 333-336. |
| [3] | 王喆, 邱林, 马贲. 番茄来源胞外囊泡样颗粒对口腔鳞状细胞癌的作用效果研究[J]. 天津医药, 2026, 54(2): 145-150. |
| [4] | 黄慧琦, 伍秋苑, 张昆, 李佩贤, 熊亚明, 叶国麟, 周丹. 川楝素联合奥拉帕尼在三阴性乳腺癌中的抗肿瘤机制研究[J]. 天津医药, 2025, 53(9): 897-902. |
| [5] | 周鹏鹏, 丁烁, 姚卫康, 罗祎. 原发性肝癌患者术前免疫因素及与病理特征的关系[J]. 天津医药, 2025, 53(9): 952-956. |
| [6] | 杨桃, 全艳, 张加孟, 谢清耘, 黄麟洲. 甲状腺结节细针穿刺细胞学联合BRAF基因检测在甲状腺良恶性肿瘤鉴别诊断中的应用价值[J]. 天津医药, 2025, 53(9): 972-975. |
| [7] | 蒋苏, 李东霞, 吕新翔, 崔艳红, 吕李婷. 基于PI3K/Akt/NF-κB信号通路探讨黄连素对特应性皮炎大鼠皮肤病理变化的治疗作用[J]. 天津医药, 2025, 53(7): 679-683. |
| [8] | 韩建存, 周谊. 川陈皮素调节FAK/AKT信号通路对喉鳞状细胞癌细胞增殖和凋亡的影响[J]. 天津医药, 2025, 53(6): 561-565. |
| [9] | 李建林, 孙思进, 王大力. Ⅰ—Ⅱ期非小细胞肺癌胸腔镜术后心肺并发症的影响因素及预测模型构建[J]. 天津医药, 2025, 53(6): 583-588. |
| [10] | 杜昀泽, 唐琼. 常规胸腔积液及血清学检测鉴别结核性及恶性胸腔积液的临床价值[J]. 天津医药, 2025, 53(6): 619-624. |
| [11] | 马春梅, 于鹏, 张其程, 杨磊, 李棣华, 谭建, 孟召伟. 异硫氰酸苄酯联合索拉非尼治疗未分化甲状腺癌机制探讨[J]. 天津医药, 2025, 53(5): 449-455. |
| [12] | 杜凌云, 王耀武, 任楠. 肾透明细胞癌中PRMT2、TRAF2与转移相关基因表达对预后的评估价值[J]. 天津医药, 2025, 53(5): 492-497. |
| [13] | 马平, 徐小明, 叶德刚. MR DWI及ADC值在鼻咽癌患者颈部淋巴结性质鉴别诊断中的应用[J]. 天津医药, 2025, 53(5): 537-541. |
| [14] | 苏红见, 张春艳, 张卫东, 韩利, 乔亚红. 鸢尾素调控EBF3/ALOX15通路影响肺腺癌细胞增殖和迁移[J]. 天津医药, 2025, 53(4): 337-342. |
| [15] | 丁明, 戎国祥, 潘忠军. 自主呼吸非气管插管硬膜外麻醉下单孔胸腔镜手术治疗非小细胞肺癌的疗效分析[J]. 天津医药, 2025, 53(4): 411-415. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||