
天津医药 ›› 2023, Vol. 51 ›› Issue (9): 928-934.doi: 10.11958/20221869
岳巾晶1( ), 曾莹1, 郭晓珮1, 董越1, 姬若楠1, 彭瑞2, 罗晓华2,△(
), 曾莹1, 郭晓珮1, 董越1, 姬若楠1, 彭瑞2, 罗晓华2,△( )
)
收稿日期:2022-11-18
修回日期:2023-03-02
出版日期:2023-09-15
发布日期:2023-09-13
通讯作者:
△E-mail:作者简介:岳巾晶(1997),女,硕士在读,主要从事子痫前期的诊断和治疗方面研究。E-mail:基金资助:
YUE Jinjing1( ), ZENG Ying1, GUO Xiaopei1, DONG Yue1, JI Ruonan1, PENG Rui2, LUO Xiaohua2,△(
), ZENG Ying1, GUO Xiaopei1, DONG Yue1, JI Ruonan1, PENG Rui2, LUO Xiaohua2,△( )
)
Received:2022-11-18
Revised:2023-03-02
Published:2023-09-15
Online:2023-09-13
Contact:
△E-mail:岳巾晶, 曾莹, 郭晓珮, 董越, 姬若楠, 彭瑞, 罗晓华. miR-155通过调控PKG1影响滋养细胞生物学功能并参与子痫前期的机制探讨[J]. 天津医药, 2023, 51(9): 928-934.
YUE Jinjing, ZENG Ying, GUO Xiaopei, DONG Yue, JI Ruonan, PENG Rui, LUO Xiaohua. MiR-155 affects the biological functions of trophoblastic cells through regulating cGMP-dependent kinase 1 and is involved in the mechanism of preeclampsia[J]. Tianjin Medical Journal, 2023, 51(9): 928-934.
摘要:
目的 探究miR-155和环磷酸鸟苷依赖性蛋白激酶1(PKG1)在子痫前期患者胎盘组织中的表达及miR-155在核因子(NF)-κB的介导下通过抑制PKG1对滋养细胞HTR-8/SVneo功能的影响。方法 收集剖宫产分娩的正常产妇(NPE组)以及子痫前期产妇(PE组)的胎盘各20个,体外培养滋养细胞HTR-8/SVneo,分为NC组、mimics组、inhibitor组、siRNA NC组、PKG1 siRNA组、siRNA+inhibitor组;用NF-κB抑制剂PDTC处理细胞,分为Control组、PDTC组、PDTC+NC组、PDTC+mimics组。采用qPCR检测胎盘和滋养细胞HTR-8/SVneo中miR-155和PKG1 mRNA的表达;Western blot检测PKG1蛋白的表达;分别采用CCK-8法、Transwell法、流式细胞术检测细胞的增殖、迁移、凋亡能力。结果 PE组胎盘组织中miR-155的表达升高,而PKG1的表达降低(P<0.05)。体外实验表明,与NC组相比,miR-155 mimics组中PKG1的表达降低,滋养细胞迁移、增殖能力减弱,凋亡能力增强,miR-155 inhibitor组中PKG1表达则升高,滋养细胞迁移、增殖能力增强,凋亡能力减弱(P<0.05);与Control组相比,PDTC组miR-155的表达降低,滋养细胞迁移、增殖能力增强,凋亡能力减弱(P<0.05)。结论 miR-155在子痫前期中表达上调,并且可在NF-κB的介导下通过下调PKG1影响滋养细胞的增殖、迁移和凋亡。
中图分类号:
| 基因名称 | 引物序列(5′→3′) | 产物大小/bp |
|---|---|---|
| U6 | 上游:CTCGCTTCGGCAGCACA | 96 |
| 下游:AACGCTTCACGAATTTGCGT | ||
| miR-155 | 上游:ACACTCCAGCTGGGTTAATGCT AATCGTGATA | 60 |
| 下游:TTAATGCTAATCGTGATAGGGG | ||
| GAPDH | 上游:TGTTCGTCATGGGTGTGAAC | 154 |
| 下游:ATGGCATGGACTGTGGTCAT | ||
| PKG1 | 上游:AACTCCACAAATGCCAGTCG | 272 |
| 下游:GCAACTGTCCTTGCCATACT |
表1 qPCR引物序列
Tab.1 Oligonucleotide primer sequences for qPCR
| 基因名称 | 引物序列(5′→3′) | 产物大小/bp |
|---|---|---|
| U6 | 上游:CTCGCTTCGGCAGCACA | 96 |
| 下游:AACGCTTCACGAATTTGCGT | ||
| miR-155 | 上游:ACACTCCAGCTGGGTTAATGCT AATCGTGATA | 60 |
| 下游:TTAATGCTAATCGTGATAGGGG | ||
| GAPDH | 上游:TGTTCGTCATGGGTGTGAAC | 154 |
| 下游:ATGGCATGGACTGTGGTCAT | ||
| PKG1 | 上游:AACTCCACAAATGCCAGTCG | 272 |
| 下游:GCAACTGTCCTTGCCATACT |
| 组别 | 年龄/岁 | BMI/(kg/m2) | SBP/mmHg |
|---|---|---|---|
| PE组 | 29.7±3.5 | 29.1±2.3 | 155.3±11.2 |
| NPE组 | 31.0±3.1 | 28.3±1.9 | 107.3±6.2 |
| t | 1.187 | 1.193 | 16.756** |
| 组别 | DPB/mmHg | 分娩孕周/周 | 胎儿出生体质量/kg |
| PE组 | 98.0±9.4 | 36.6±2.1 | 2.6±0.7 |
| NPE组 | 67.8±6.3 | 37.0±1.8 | 3.0±0.5 |
| t | 11.957** | 0.730 | 3.235* |
表2 2组产妇一般资料比较(n=20,$\bar{x}\pm s$)
Tab.2 Comparison of clinical characteristics between the two groups of patients
| 组别 | 年龄/岁 | BMI/(kg/m2) | SBP/mmHg |
|---|---|---|---|
| PE组 | 29.7±3.5 | 29.1±2.3 | 155.3±11.2 |
| NPE组 | 31.0±3.1 | 28.3±1.9 | 107.3±6.2 |
| t | 1.187 | 1.193 | 16.756** |
| 组别 | DPB/mmHg | 分娩孕周/周 | 胎儿出生体质量/kg |
| PE组 | 98.0±9.4 | 36.6±2.1 | 2.6±0.7 |
| NPE组 | 67.8±6.3 | 37.0±1.8 | 3.0±0.5 |
| t | 11.957** | 0.730 | 3.235* |
| 组别 | miR-155 | PKG1 |
|---|---|---|
| PE组 | 4.41±2.06 | 1.52±0.90 |
| NPE组 | 2.07±0.61 | 4.04±2.03 |
| t | 4.782** | 5.076** |
表3 2组胎盘组织中miR-155及PKG1 mRNA表达水平比较(n=20,$\bar{x}\pm s$)
Tab.3 Comparison of expression levels of miR-155 and PKG1 mRNA in placental tissue between the two groups
| 组别 | miR-155 | PKG1 |
|---|---|---|
| PE组 | 4.41±2.06 | 1.52±0.90 |
| NPE组 | 2.07±0.61 | 4.04±2.03 |
| t | 4.782** | 5.076** |
| 组别 | miR-155 | PKG1 mRNA | PKG1蛋白 |
|---|---|---|---|
| NC组 | 1.00±0.07 | 1.00±0.23 | 0.49±0.10 |
| miR-155 mimics组 | 12.41±1.02a | 0.41±0.03a | 0.13±0.01 a |
| miR-155 inhibitor组 | 0.29±0.02a | 2.76±0.31a | 1.19±0.07a |
| F | 400.478** | 135.734** | 185.053** |
表4 miR-155 mimics和miR-155 inhibitor处理后各组miR-155、PKG1 mRNA和蛋白水平比较 (n=3,$\bar{x}\pm s$)
Tab.4 Comparison of miR-155 and PKG1 mRNA expression levels after treatment with miR-155 mimics and miR-155 inhibitor between the three groups
| 组别 | miR-155 | PKG1 mRNA | PKG1蛋白 |
|---|---|---|---|
| NC组 | 1.00±0.07 | 1.00±0.23 | 0.49±0.10 |
| miR-155 mimics组 | 12.41±1.02a | 0.41±0.03a | 0.13±0.01 a |
| miR-155 inhibitor组 | 0.29±0.02a | 2.76±0.31a | 1.19±0.07a |
| F | 400.478** | 135.734** | 185.053** |
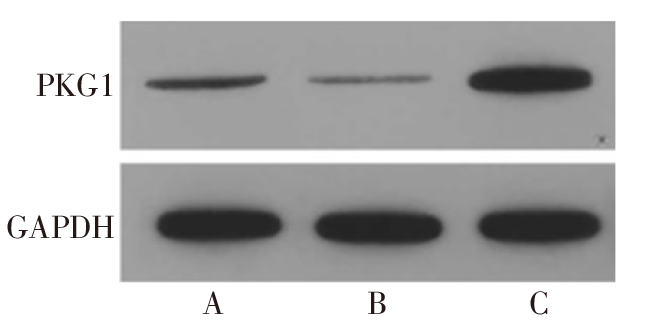
图2 Western blot检测各组滋养细胞HTR-8/SVneo中PKG1蛋白的表达 A:NC组;B:miR-155 mimics组;C:miR-155 inhibitor组。
Fig.2 Expression of PKG1 protein in each group of trophoblast HTR-8/SVneo detected by Western blot assay
| 组别 | 增殖能力(A450) | ||
|---|---|---|---|
| 24 h | 48 h | 72 h | |
| NC组 miR-155 mimics组 miR-155 inhibitor组 | 0.26±0.02 0.25±0.01a 0.29±0.01a | 0.43±0.21 0.32±0.03a 0.50±0.03a | 0.64±0.19 0.46±0.02a 0.87±0.04a |
| F | 6.048* | 37.327** | 160.188** |
| 组别 | 迁移细胞数/(个/视野) | 凋亡率/% | |
| NC组 miR-155 mimics组 miR-155 inhibitor组 | 124.67±11.37 64.33±10.02a 205.33±8.02a | 11.58±1.14 17.56±1.44a 5.80±0.54a | |
| F | 153.205** | 85.114** | |
表5 miR-155 mimics和miR-155 inhibitor处理后各组细胞增殖、迁移及凋亡水平的比较(n=3,$\bar{x}\pm s$)
Tab.5 Comparison of cell proliferation, migration and apoptosis after treatment with miR-155 mimics and miR-155 inhibitor between the three groups
| 组别 | 增殖能力(A450) | ||
|---|---|---|---|
| 24 h | 48 h | 72 h | |
| NC组 miR-155 mimics组 miR-155 inhibitor组 | 0.26±0.02 0.25±0.01a 0.29±0.01a | 0.43±0.21 0.32±0.03a 0.50±0.03a | 0.64±0.19 0.46±0.02a 0.87±0.04a |
| F | 6.048* | 37.327** | 160.188** |
| 组别 | 迁移细胞数/(个/视野) | 凋亡率/% | |
| NC组 miR-155 mimics组 miR-155 inhibitor组 | 124.67±11.37 64.33±10.02a 205.33±8.02a | 11.58±1.14 17.56±1.44a 5.80±0.54a | |
| F | 153.205** | 85.114** | |
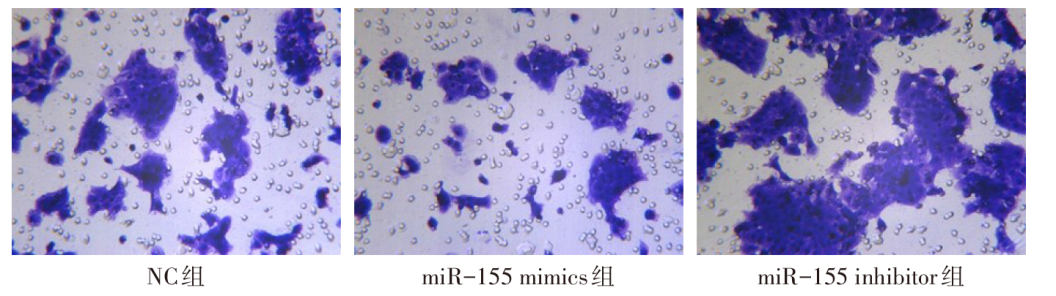
图3 miR-155 mimics和miR-155 inhibitor处理后各组细胞迁移情况(×200)
Fig.3 Cell migration of each group after treatment with miR-155 mimics and miR-155 inhibitor (×200)
| 组别 | miR-155 | PKG1 mRNA | PKG1蛋白 |
|---|---|---|---|
| NC组 | 0.99±0.01 | 1.02±0.04 | 0.21±0.04 |
| inhibitor组 | 0.32±0.04a | 3.06±0.19a | 0.83±0.03a |
| siRNA NC组 | 1.07±0.09 | 1.00±0.03 | 0.25±0.05 |
| PKG1 siRNA组 | 1.04±0.08 | 0.25±0.04 | 0.10±0.00 |
| siRNA+inhibitor组 | 0.65±0.07b | 0.64±0.08b | 0.34±0.00b |
| F | 10.933* | 12.000* | 67.230** |
表6 抑制PKG1的表达后各组miR-155、PKG1 mRNA和蛋白水平比较(n=3,$\bar{x}\pm s$)
Tab.6 Comparison of miR-155, PKG1 mRNA and protein levels after inhibiting the expression of PKG1 between the five groups
| 组别 | miR-155 | PKG1 mRNA | PKG1蛋白 |
|---|---|---|---|
| NC组 | 0.99±0.01 | 1.02±0.04 | 0.21±0.04 |
| inhibitor组 | 0.32±0.04a | 3.06±0.19a | 0.83±0.03a |
| siRNA NC组 | 1.07±0.09 | 1.00±0.03 | 0.25±0.05 |
| PKG1 siRNA组 | 1.04±0.08 | 0.25±0.04 | 0.10±0.00 |
| siRNA+inhibitor组 | 0.65±0.07b | 0.64±0.08b | 0.34±0.00b |
| F | 10.933* | 12.000* | 67.230** |
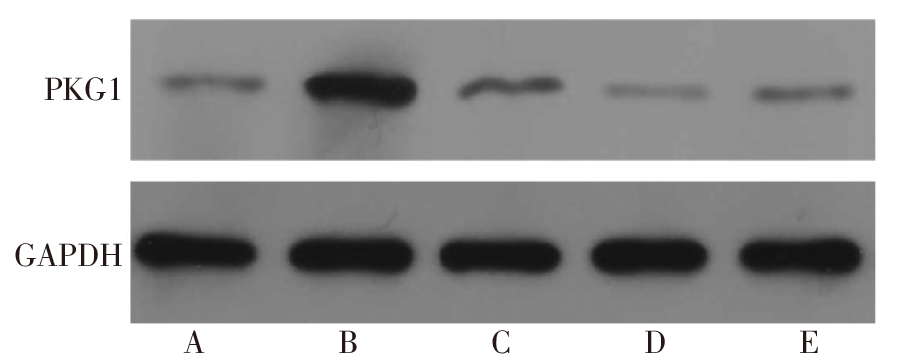
图5 Western blot检测各组滋养细胞HTR-8/SVneo中PKG1蛋白的表达 A:NC组;B:inhibitor组;C:siRNA NC组;D:PKG1 siRNA组;E:siRNA+inhibitor组。
Fig.5 Expression of PKG1 protein in each group of trophoblast HTR-8/SVneo detected by Western blot assay
| 组别 | miR-155 mRNA | PKG1 mRNA | PKG1蛋白 |
|---|---|---|---|
| Control组 | 1.00±0.03 | 1.01±0.13 | 0.35±0.02 |
| PDTC组 | 0.56±0.05a | 3.24±0.23a | 1.00±0.04a |
| PDTC+NC组 | 0.57±0.01 | 3.13±0.25 | 1.01±0.05 |
| PDTC+ mimics组 | 10.11±0.28b | 0.50±0.01b | 0.11±0.03b |
| F | 3 233.927** | 182.535** | 311.788** |
表7 抑制NF-κB表达后各组细胞中miR-155、PKG1 mRNA和蛋白表达水平比较(n=3,$\bar{x}\pm s$)
Tab.7 Comparison of miR-155 mimics and miR-155 mRNA expression levels after inhibiting the expression of NF-κB between the four groups
| 组别 | miR-155 mRNA | PKG1 mRNA | PKG1蛋白 |
|---|---|---|---|
| Control组 | 1.00±0.03 | 1.01±0.13 | 0.35±0.02 |
| PDTC组 | 0.56±0.05a | 3.24±0.23a | 1.00±0.04a |
| PDTC+NC组 | 0.57±0.01 | 3.13±0.25 | 1.01±0.05 |
| PDTC+ mimics组 | 10.11±0.28b | 0.50±0.01b | 0.11±0.03b |
| F | 3 233.927** | 182.535** | 311.788** |

图6 Western blot检测各组滋养细胞HTR-8/SVneo中PKG1蛋白的表达 A:Control组;B:PDTC组;C:PDTC+NC组;D:PDTC+ mimics组。
Fig.6 Expression of PKG1 protein in each group of trophoblast HTR-8/SVneo detected by Western blot assay
| 组别 | 增殖能力(A450) | ||
|---|---|---|---|
| 24 h | 48 h | 72 h | |
| Control组 | 0.26±0.01 | 0.42±0.04 | 0.63±0.02 |
| PDTC组 | 0.29±0.02a | 0.59±0.04a | 0.93±0.04a |
| PDTC+NC组 | 0.30±0.03 | 0.53±0.03 | 0.90±0.06 |
| PDTC+mimics组 | 0.24±0.02b | 0.33±0.01b | 0.45±0.03b |
| F | 6.267* | 38.309** | 104.970** |
| 组别 | 迁移细胞数/(个/视野) | 凋亡率/% | |
| Control组 | 116.00±5.29 | 10.13±0.96 | |
| PDTC组 | 169.67±8.62a | 6.25±1.06a | |
| PDTC+NC组 | 156.67±4.17 | 6.59±1.09 | |
| PDTC+mimics组 | 64.00±6.25b | 15.56±0.06b | |
| F | 171.173** | 69.012** | |
表8 抑制NF-κB表达后各组细胞增殖、迁移及凋亡率比较(n=3,$\bar{x}\pm s$)
Tab.8 Comparison of cell proliferation, migration and apoptosis after inhibiting the expression of NF-κB between the four groups
| 组别 | 增殖能力(A450) | ||
|---|---|---|---|
| 24 h | 48 h | 72 h | |
| Control组 | 0.26±0.01 | 0.42±0.04 | 0.63±0.02 |
| PDTC组 | 0.29±0.02a | 0.59±0.04a | 0.93±0.04a |
| PDTC+NC组 | 0.30±0.03 | 0.53±0.03 | 0.90±0.06 |
| PDTC+mimics组 | 0.24±0.02b | 0.33±0.01b | 0.45±0.03b |
| F | 6.267* | 38.309** | 104.970** |
| 组别 | 迁移细胞数/(个/视野) | 凋亡率/% | |
| Control组 | 116.00±5.29 | 10.13±0.96 | |
| PDTC组 | 169.67±8.62a | 6.25±1.06a | |
| PDTC+NC组 | 156.67±4.17 | 6.59±1.09 | |
| PDTC+mimics组 | 64.00±6.25b | 15.56±0.06b | |
| F | 171.173** | 69.012** | |
| [1] | IVES C W, SINKEY R, RAJAPREYAR I, et al. Preeclampsia-pathophysiology and clinical presentations:JACC state-of-the-art review[J]. J Am Coll Cardiol, 2020, 76(14):1690-1702. doi:10.1016/j.jacc.2020.08.014. |
| [2] | ALFAIFI A A, HEYDER R S, BIELSKI E R, et al. Megalin-targeting liposomes for placental drug delivery[J]. J Control Release, 2020, 324:366-378. doi:10.1016/j.jconrel.2020.05.033. |
| [3] | ZHOU Y, WANG J, WANG L, et al. Effect of compound danshen injection combined with magnesium sulfate on oxidative stress,TNF-alpha,NO,and therapeutic efficacy in severe preeclampsia[J]. Comput Intell Neurosci, 2022, 2022:9789066. doi:10.3390/biom10060953. |
| [4] | SHIRVANI S O, SCHERR J, KAYVANPOUR E, et al. Marathon-induced cardiac strain as model for the evaluation of diagnostic microRNAs for acute myocardial infarction[J]. J Clin Med, 2021, 11(1):5. doi:10.3390/jcm11010005. |
| [5] | KOLKOVA Z, HOLUBEKOVA V, GRENDAR M, et al. Association of circulating miRNA expression with preeclampsia,its onset,and severity[J]. Diagnostics (Basel), 2021, 11(3):476. doi:10.3390/diagnostics11030476. |
| [6] | LUO X, PAN C, GUO X, et al. Methylation mediated silencing of miR-155 suppresses the development of preeclampsia in vitro and in vivo by targeting FOXO3[J]. Mediators Inflamm, 2022, 2022:4250621. doi:10.1155/2022/4250621. |
| [7] | CHOI S, PARK M, KIM J, et al. TNF-alpha elicits phenotypic and functional alterations of vascular smooth muscle cells by miR-155-5p-dependent down-regulation of cGMP-dependent kinase 1[J]. J Biol Chem, 2018, 293(38):14812-14822. doi:10.1074/jbc.RA118.004220. |
| [8] | 中华医学会妇产科学分会妊娠期高血压疾病学组. 妊娠期高血压疾病诊治指南(2020)[J]. 中华妇产科杂志, 2020, 55(4):227-238. |
| Group of Hypertensive Disorders of Pregnancy,Obstetrics and Gynecology Branch of Chinese Medical Association. Guidelines for the diagnosis and treatment of hypertensive disorders during pregnancy(2020)[J]. Chin J Obstetr Gynecol, 2020, 55(4):227-238. doi:10.3760/cma.j.cn112141-20200114-00039. | |
| [9] | LI H, OUYANG Y, SADOVSKY E, et al. Unique microRNA signals in plasma exosomes from pregnancies complicated by preeclampsia[J]. Hypertension, 2020, 75(3):762-771. doi:10.1161/HYPERTENSIONAHA.119.14081. |
| [10] | ABDELZAHER W Y, MOSTAFA-HEDEAB G, BAHAA H A, et al. Leukotriene receptor antagonist,montelukast ameliorates L-NAME-induced pre-eclampsia in rats through suppressing the IL-6/Jak2/STAT3 signaling pathway[J]. Pharmaceuticals(Basel), 2022, 15(8):914. doi:10.3390/ph15080914. |
| [11] | LIU X, WANG W, BAI Y, et al. Identification of a genome-wide serum microRNA expression profile as potential noninvasive biomarkers for chronic kidney disease using next-generation sequencing[J]. J Int Med Res, 2020, 48(12):1220769033. doi:10.1177/0300060520969481. |
| [12] | CAI M, KOLLURU G K, AHMED A. Small molecule,big prospects:microRNA in pregnancy and its complications[J]. J Pregnancy, 2017, 201:6972732. doi:10.1155/2017/6972732. |
| [13] | HEMMATZADEH M, SHOMALI N, YOUSEFZADEH Y, et al. MicroRNAs:small molecules with a large impact on pre-eclampsia[J]. J Cell Physiol, 2020, 235(4):3235-3248. doi:10.1002/jcp.29286. |
| [14] | WU H Y, LIU K, ZHANG J L. LINC00240/miR-155 axis regulates function of trophoblasts and M2 macrophage polarization via modulating oxidative stress-induced pyroptosis in preeclampsia[J]. Mol Med, 2022, 28(1):119. doi:10.1186/s10020-022-00531-3. |
| [15] | EVORA P, SOARES R, BASSETTO S, et al. After thirty years,we still cannot understand why methylene blue is not a reference to treat vasoplegic syndrome in cardiac surgery[J]. Braz J Cardiovasc Surg, 2021, 36(3):406-411. doi:10.21470/1678-9741-2021-0955. |
| [16] | ITO H, MORISHITA R, NAGATA K I. Functions of rhotekin,an effector of rho GTPase,and its binding partners in mammals[J]. Int J Mol Sci, 2018, 19(7):2121. doi:10.3390/ijms19072121. |
| [17] | RAMDANI G, SCHALL N, KALYANARAMAN H, et al. cGMP-dependent protein kinase-2 regulates bone mass and prevents diabetic bone loss[J]. J Endocrinol, 2018, 238(3):203-219. doi:10.1530/JOE-18-0286. |
| [18] | BISWAS S, KOJONAZAROV B, HADZIC S, et al. IRAG1 deficient mice develop PKG1beta dependent pulmonary hypertension[J]. Cells, 2020, 9(10):2280. doi:10.3390/cells9102280. |
| [19] | FERNANDO I P S, KIRINDAGE K G I S, JAYASINGHE A M K, et al. Hot water extract of Sasa borealis (hack.) makino & shibata abate hydrogen peroxide-induced oxidative stress and apoptosis in kidney epithelial cells[J]. Antioxidants(Basel), 2022, 11(5):1013. doi:10.3390/antiox11051013. |
| [20] | ZHAO L, LI Y, YAO D, et al. Pharmacological basis for use of a novel compound in hyperuricemia:anti-hyperuricemic and anti-inflammatory effects[J]. Front Pharmacol, 2021, 12:772504. doi:10.3389/fphar.2021.772504. |
| [21] | STAFF A C. The two-stage placental model of preeclampsia:an update[J]. J Reprod Immunol, 2019:134-135. doi:10.1016/j.jri.2019.07.004. |
| [22] | WANG Y, LI B, ZHAO Y. Inflammation in preeclampsia:genetic biomarkers, mechanisms,and therapeutic strategies[J]. Front Immunol, 2022, 13:883404. doi:10.3389/fimmu.2022.883404. |
| [23] | SOCHA M W, MALINOWSKI B, PUK O, et al. The role of NF-κB in uterine spiral arteries remodeling,insight into the cornerstone of preeclampsia[J]. Int J Mol Sci, 2021, 22(2):704. doi:10.3390/ijms22020704. |
| [24] | MANN M, MEHTA A, ZHAO J L, et al. An NF-κB-microRNA regulatory network tunes macrophage inflammatory responses[J]. Nat Commun, 2017, 8(1):851. doi:10.1038/s41467-017-00972-z. |
| [1] | 姜天佑, 李敏, 孙碧文, 李越洋, 邢丽静, 田晨. Let-7b诱导白血病相关巨噬细胞复极抑制AML的发展[J]. 天津医药, 2026, 54(3): 225-231. |
| [2] | 杨晓芳, 贾新燕, 丰文君. miR-181a-5p通过HMGB1/NF-κB信号通路调控狼疮性肾炎小鼠肾小球系膜细胞增殖和凋亡[J]. 天津医药, 2026, 54(3): 232-237. |
| [3] | 张婧, 魏玉英, 宁海虹, 韦红梅, 王嘉玮, 曹薇, 吴宾. DUSP9在2型糖尿病心肌病小鼠心肌损伤中的保护作用及其机制[J]. 天津医药, 2026, 54(3): 238-244. |
| [4] | 王喆, 邱林, 马贲. 番茄来源胞外囊泡样颗粒对口腔鳞状细胞癌的作用效果研究[J]. 天津医药, 2026, 54(2): 145-150. |
| [5] | 李志伟, 张会超, 杨凤鸣, 曾垂义. 基于miR-144-3p/MAPK1通路探讨红参总皂苷对扩张型心肌病小鼠心肌细胞凋亡的影响[J]. 天津医药, 2026, 54(1): 23-29. |
| [6] | 赵兰君, 李良惠, 马馨, 巩娇娇, 郑臣辉, 石琳. 穿心莲内酯调控STAT3/GPX4通路对骨髓瘤细胞增殖和凋亡的影响[J]. 天津医药, 2026, 54(1): 8-13. |
| [7] | 孔翠文, 路延双, 孙丽萍, 于芬芬. LncRNA SNHG14靶向miR-30a-5p对高糖诱导的足细胞损伤的影响[J]. 天津医药, 2025, 53(9): 903-909. |
| [8] | 刘虹, 张玥玥, 王一琳, 王彩丽, 王晓敏, 毛敏, 李燕. MicroRNA-34a通过调控Wnt途径影响慢性淋巴细胞白血病进展的机制探讨[J]. 天津医药, 2025, 53(8): 785-790. |
| [9] | 万艳波, 刘明, 王勇. 秦皮甲素调节HMGB1/RAGE信号通路对缺氧/复氧诱导的心肌细胞损伤的影响[J]. 天津医药, 2025, 53(8): 796-801. |
| [10] | 刘海威, 杨洁, 王力, 蒙诗波, 唐旭松, 刘成仁, 王永旺. 木犀草素通过NFE2L2/x-CT/GPX4信号轴调控ROS水平抑制胶质母细胞瘤[J]. 天津医药, 2025, 53(7): 673-678. |
| [11] | 蒋苏, 李东霞, 吕新翔, 崔艳红, 吕李婷. 基于PI3K/Akt/NF-κB信号通路探讨黄连素对特应性皮炎大鼠皮肤病理变化的治疗作用[J]. 天津医药, 2025, 53(7): 679-683. |
| [12] | 韩建存, 周谊. 川陈皮素调节FAK/AKT信号通路对喉鳞状细胞癌细胞增殖和凋亡的影响[J]. 天津医药, 2025, 53(6): 561-565. |
| [13] | 马春梅, 于鹏, 张其程, 杨磊, 李棣华, 谭建, 孟召伟. 异硫氰酸苄酯联合索拉非尼治疗未分化甲状腺癌机制探讨[J]. 天津医药, 2025, 53(5): 449-455. |
| [14] | 余朝霞, 马贲, 邱林, 高倩, 尼娜. 基于网络药理学和实验验证探究鲍式层孔菌多酚的抗头颈鳞癌机制[J]. 天津医药, 2025, 53(5): 456-461. |
| [15] | 李晨, 李占恩, 苏宏伟, 侯彩云, 董少文. KRT17调节Wnt/β-catenin信号通路对膀胱癌细胞增殖、凋亡及上皮间质转化的影响[J]. 天津医药, 2025, 53(5): 462-467. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||