
天津医药 ›› 2022, Vol. 50 ›› Issue (10): 1014-1019.doi: 10.11958/20220649
郭玉静( ), 胡英, 龙启福, 许玉珍, 李积东, 永胜(
), 胡英, 龙启福, 许玉珍, 李积东, 永胜( ),△
),△
收稿日期:2022-04-27
修回日期:2022-06-07
出版日期:2022-10-15
发布日期:2022-10-20
通讯作者:
△永胜
E-mail:ly47757@163.com;yongsheng@qhu.edu.cn
作者简介:郭玉静(1998),女,硕士在读,主要从事低氧对免疫功能改变及适应机制方面研究。E-mail: 基金资助:
GUO Yujing( ), HU Ying, LONG Qifu, XU Yuzhen, LI Jidong, YONG Sheng(
), HU Ying, LONG Qifu, XU Yuzhen, LI Jidong, YONG Sheng( ),△
),△
Received:2022-04-27
Revised:2022-06-07
Published:2022-10-15
Online:2022-10-20
Contact:
△YONG Sheng
E-mail:ly47757@163.com;yongsheng@qhu.edu.cn
郭玉静, 胡英, 龙启福, 许玉珍, 李积东, 永胜. 低氧暴露对小鼠脾淋巴细胞增殖和凋亡的影响[J]. 天津医药, 2022, 50(10): 1014-1019.
GUO Yujing, HU Ying, LONG Qifu, XU Yuzhen, LI Jidong, YONG Sheng. Effects of hypoxia exposure on proliferation and apoptosis of mouse spleen lymphocytes[J]. Tianjin Medical Journal, 2022, 50(10): 1014-1019.
摘要:
目的 研究低氧暴露诱导淋巴细胞数量减少与细胞增殖和凋亡的关系。方法 从C57BL/6小鼠脾中分离淋巴细胞,将淋巴细胞分别在低氧(1%O2)和常氧(21%O2)环境中培养12、24、48 h。用流式细胞术检测T、B淋巴细胞数量;羧基荧光素乙酰乙酸和AnnexinⅤ-FITC/PI方法分别检测淋巴细胞的增殖和凋亡;扫描电镜观察淋巴细胞形态;实时荧光定量逆转录PCR和Western blot检测凋亡相关因子鼠源B细胞淋巴瘤/白血病-2(bcl-2)、bcl-2同源拮抗剂-杀伤蛋白(Bak)和胱天蛋白酶-3(caspase-3)的mRNA和蛋白表达水平。结果 低氧暴露12、24、48h,可降低T、B淋巴细胞数量,抑制淋巴细胞增殖,促进细胞凋亡;扫描电镜观察显示,同一时间下,低氧组淋巴细胞更早出现凋亡特征形态学变化;低氧暴露12、24、48 h,淋巴细胞Bak和caspase-3的mRNA和蛋白表达水平均上调,而bcl-2的mRNA和蛋白表达水平均下调。结论 低氧暴露通过抑制淋巴细胞增殖、促进凋亡,从而介导淋巴细胞数量下降。
中图分类号:
| 基因 名称 | 引物序列(5′→3′) | 产物大小 (bp) |
|---|---|---|
| β-actin | 上游:CATCCGTAAAGACCTCTATGCCAAC | 171 |
| 下游:ATGGAGCCACCGATCCACA | ||
| Bak | 上游:GGTCTTTCGAAGCTACGTTTTT | 243 |
| 下游:ATCTTGGTGAAGAGTTCGTAGG | ||
| bcl-2 | 上游:GATGACTTCTCTCGTCGCTAC | 156 |
| 下游:GAACTCAAAGAAGGCCACAATC | ||
| caspase-3 | 上游:GGCCTGAAATACCAAGTCAGGAA | 128 |
| 下游:CCATGGCTTAGAATCACACACACA |
表1 qPCR引物序列
Tab. 1 Sequence of the primers in qPCR
| 基因 名称 | 引物序列(5′→3′) | 产物大小 (bp) |
|---|---|---|
| β-actin | 上游:CATCCGTAAAGACCTCTATGCCAAC | 171 |
| 下游:ATGGAGCCACCGATCCACA | ||
| Bak | 上游:GGTCTTTCGAAGCTACGTTTTT | 243 |
| 下游:ATCTTGGTGAAGAGTTCGTAGG | ||
| bcl-2 | 上游:GATGACTTCTCTCGTCGCTAC | 156 |
| 下游:GAACTCAAAGAAGGCCACAATC | ||
| caspase-3 | 上游:GGCCTGAAATACCAAGTCAGGAA | 128 |
| 下游:CCATGGCTTAGAATCACACACACA |
| 组别 | 12 h | 24 h | 48 h | |||
|---|---|---|---|---|---|---|
| T淋巴细胞 | B淋巴细胞 | T淋巴细胞 | B淋巴细胞 | T淋巴细胞 | B淋巴细胞 | |
| 常氧组 | 40.17±1.90 | 33.47±2.38 | 32.65±4.60 | 27.96±1.50 | 26.30±1.11 | 24.62±2.88 |
| 低氧组 | 32.26±3.30 | 27.42±2.73 | 23.77±2.61 | 23.39±2.31 | 23.16±1.33 | 18.71±1.31 |
| t | 3.603* | 2.895* | 2.904* | 2.873* | 3.140* | 3.229* |
表2 2组不同时间T、B淋巴细胞百分比比较 (n=3,%,$\bar{x} \pm s$)
Tab. 2 Comparison of the percentages of T and B lymphocytes at different time points between the two groups
| 组别 | 12 h | 24 h | 48 h | |||
|---|---|---|---|---|---|---|
| T淋巴细胞 | B淋巴细胞 | T淋巴细胞 | B淋巴细胞 | T淋巴细胞 | B淋巴细胞 | |
| 常氧组 | 40.17±1.90 | 33.47±2.38 | 32.65±4.60 | 27.96±1.50 | 26.30±1.11 | 24.62±2.88 |
| 低氧组 | 32.26±3.30 | 27.42±2.73 | 23.77±2.61 | 23.39±2.31 | 23.16±1.33 | 18.71±1.31 |
| t | 3.603* | 2.895* | 2.904* | 2.873* | 3.140* | 3.229* |
| 组别 | 12 h | 24 h | 48 h |
|---|---|---|---|
| 常氧组 | 31.56±1.63 | 38.50±0.31 | 43.32±1.35 |
| 低氧组 | 27.31±2.05 | 34.74±0.82 | 40.66±0.49 |
| t | 4.749** | 11.324** | 5.566** |
表3 2组不同时间淋巴细胞增殖率比较 (n=3,%,$\bar{x} \pm s$)
Tab. 3 Comparison of lymphocyte proliferation rates at different time points between two groups
| 组别 | 12 h | 24 h | 48 h |
|---|---|---|---|
| 常氧组 | 31.56±1.63 | 38.50±0.31 | 43.32±1.35 |
| 低氧组 | 27.31±2.05 | 34.74±0.82 | 40.66±0.49 |
| t | 4.749** | 11.324** | 5.566** |
| 组别 | 12 h | 24 h | 48 h |
|---|---|---|---|
| 常氧组 | 17.05±1.33 | 30.66±3.11 | 82.01±1.66 |
| 低氧组 | 22.28±1.13 | 36.34±1.64 | 86.99±1.42 |
| t | 5.446** | 3.172* | 6.033** |
表4 2组不同时间淋巴细胞凋亡率比较 (n=3,%,$\bar{x} \pm s$)
Tab. 4 Comparison of apoptosis rate of lymphocyte at different time points between the two groups
| 组别 | 12 h | 24 h | 48 h |
|---|---|---|---|
| 常氧组 | 17.05±1.33 | 30.66±3.11 | 82.01±1.66 |
| 低氧组 | 22.28±1.13 | 36.34±1.64 | 86.99±1.42 |
| t | 5.446** | 3.172* | 6.033** |
| 组别 | 12 h | 24 h | 48 h | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Bak | bcl-2 | caspase-3 | Bak | bcl-2 | caspase-3 | Bak | bcl-2 | caspase-3 | |
| 常氧组 | 0.98±0.12 | 1.00±0.10 | 0.96±0.08 | 1.01±0.36 | 1.10±0.30 | 0.94±0.08 | 0.95±0.49 | 1.16±0.47 | 0.98±0.16 |
| 低氧组 | 1.11±0.18 | 0.66±0.21 | 1.25±0.35 | 1.43±0.28 | 0.52±0.22 | 1.15±0.19 | 1.44±0.42 | 0.74±0.39 | 2.21±0.56 |
| t | 2.248* | 4.527** | 2.171* | 2.724* | 4.440** | 2.532* | 2.721* | 2.516* | 5.973** |
表5 2组低氧暴露不同时间淋巴细胞Bak、bcl-2、caspase-3 mRNA表达水平比较 (n=3,$\bar{x} \pm s$)
Tab. 5 Comparison of mRNA expression levels of Bak, bcl-2 and caspase-3 in lymphocytes at different time points between the two groups
| 组别 | 12 h | 24 h | 48 h | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Bak | bcl-2 | caspase-3 | Bak | bcl-2 | caspase-3 | Bak | bcl-2 | caspase-3 | |
| 常氧组 | 0.98±0.12 | 1.00±0.10 | 0.96±0.08 | 1.01±0.36 | 1.10±0.30 | 0.94±0.08 | 0.95±0.49 | 1.16±0.47 | 0.98±0.16 |
| 低氧组 | 1.11±0.18 | 0.66±0.21 | 1.25±0.35 | 1.43±0.28 | 0.52±0.22 | 1.15±0.19 | 1.44±0.42 | 0.74±0.39 | 2.21±0.56 |
| t | 2.248* | 4.527** | 2.171* | 2.724* | 4.440** | 2.532* | 2.721* | 2.516* | 5.973** |
| 组别 | 12 h | 24 h | 48 h | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Bak | bcl-2 | caspase-3 | Bak | bcl-2 | caspase-3 | Bak | bcl-2 | caspase-3 | |
| 常氧组 | 0.31±0.11 | 0.62±0.06 | 0.45±0.08 | 0.23±0.15 | 0.90±0.14 | 0.71±0.06 | 0.37±0.13 | 0.36±0.04 | 0.36±0.03 |
| 低氧组 | 0.54±0.09 | 0.44±0.09 | 0.72±0.15 | 0.48±0.03 | 0.50±0.17 | 0.94±0.13 | 0.73±0.17 | 0.23±0.02 | 0.66±0.10 |
| t | 2.828* | 2.819* | 2.794* | 2.799* | 3.117* | 2.787* | 2.867* | 4.658** | 4.950** |
表6 2组低氧暴露不同时间淋巴细胞Bak、bcl-2、caspase-3蛋白表达水平比较 (n=3,$\bar{x} \pm s$)
Tab. 6 Comparison of Bak, bcl-2 and caspase-3 protein expression levels in lymphocytes at different time points between the two groups of mice
| 组别 | 12 h | 24 h | 48 h | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Bak | bcl-2 | caspase-3 | Bak | bcl-2 | caspase-3 | Bak | bcl-2 | caspase-3 | |
| 常氧组 | 0.31±0.11 | 0.62±0.06 | 0.45±0.08 | 0.23±0.15 | 0.90±0.14 | 0.71±0.06 | 0.37±0.13 | 0.36±0.04 | 0.36±0.03 |
| 低氧组 | 0.54±0.09 | 0.44±0.09 | 0.72±0.15 | 0.48±0.03 | 0.50±0.17 | 0.94±0.13 | 0.73±0.17 | 0.23±0.02 | 0.66±0.10 |
| t | 2.828* | 2.819* | 2.794* | 2.799* | 3.117* | 2.787* | 2.867* | 4.658** | 4.950** |
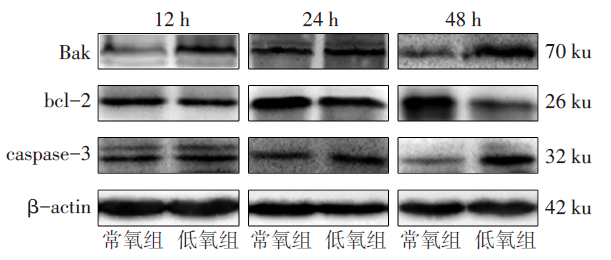
图4 低氧暴露不同时间淋巴细胞Bak、bcl-2、caspase-3的蛋白表达水平
Fig.4 Protein expression levels of Bak, bcl-2 and caspase-3 in lymphocytes at different time points between the two groups of mice
| [1] | ZHANG T, SUO C, ZHENG C, et al. Hypoxia and metabolism in metastasis[J]. Adv Exp Med Biol, 2019, 1136:87-95. doi: 10.1007/978-3-030-12734-3_6. |
| [2] | LUNDEBERG J, FEINER J R, SCHOBER A, et al. Increased cytokines at high altitude:Lack of effect of ibuprofen on acute mountain sickness,physiological variables,or cytokine levels[J]. High Alt Med Biol, 2018, 19(3):249-258. doi: 10.1089/ham.2017.0144. |
| [3] | 王新, 孙蓓, 刘芳, 等. 间歇低氧下小鼠血管内皮功能障碍机制的研究[J]. 天津医药, 2017, 45(2):160-163. |
| WANG X, SUN B, LIU F, et al. Studies of the mechanism of endothelial dysfunction in rats under intermittent hypoxia[J]. Tianjin Med J, 2017, 45(2):160-163. doi: 10.11958/20161305. | |
| [4] | WANG J, JIANG R, TAN Y, et al. Human pulmonary artery smooth muscle cell dysfunction is regulated by miR-509-5p in hypoxic environment[J]. Cell Cycle, 2022, 21(11):1212-1221. doi: 10.1080/15384101.2022.2044147. |
| [5] | NOMAN M Z, DESANTIS G, JANJI B, et al. PD-L1 is a novel direct target of HIF-1α,and its blockade under hypoxia enhanced MDSC-mediated T cell activation[J]. J Exp Med, 2014, 211(5):781-790. doi: 10.1084/jem.20131916. |
| [6] | BURROWS N, BASHFORD-ROGERS R J M, BHUTE V J, et al. Dynamic regulation of hypoxia-inducible factor-1α activity is essential for normal B cell development[J]. Nat Immunol, 2020, 21(11):1408-1420. doi: 10.1038/s41590-020-0772-8. |
| [7] | JIANG X, TIAN W, KIM D, et al. Hypoxia and hypoxia-inducible factors in lymphedema[J]. Front Pharmacol, 2022, 13:851057. doi: 10.3389/fphar.2022.851057. |
| [8] | 张晓娜, 李积东, 胡方杰, 等. 高原低氧暴露小鼠脾脏T淋巴细胞数量减少且免疫活性下降[J]. 细胞与分子免疫学杂志, 2017, 33(2):164-167. |
| ZHANG X N, LI J D, HU F J, et al. Decreased number and immune activity of splenic T lymphocytes in mice exposed to hypoxia at high altitude[J]. Chinese Journal of Cellular and Molecular Immunology, 2017, 33(2):164-167. doi: 10.13423/j.cnki.cjcmi.008007. | |
| [9] | XIE L, LIN Y, DENG Y, et al. The effect of SARS-CoV-2 on the spleen and T lymphocytes[J]. Viral Immunol, 2021, 34(6):416-420. doi: 10.1089/vim.2020.0320. |
| [10] | HU F, LIU H, XU L, et al. Hypoxia-inducible factor-1α perpetuates synovial fibroblast interactions with T cells and B cells in rheumatoid arthritis[J]. Eur J Immunol, 2016, 46(3):742-751. doi: 10.1002/eji.201545784. |
| [11] | LIU Y N, YANG J F, HUANG D J, et al. Hypoxia induces mitochondrial defect that promotes T cell exhaustion in tumor microenvironment through MYC-regulated pathways[J]. Front Immunol, 2020, 11:1906. doi: 10.3389/fimmu.2020.01906. |
| [12] | BYRNES J R, WEEKS A M, SHIFRUT E, et al. Hypoxia is a dominant remodeler of the effector T cell surface proteome relative to activation and regulatory T cell suppression[J]. Mol Cell Proteomics, 2022, 21(4):100217. doi: 10.1016/j.mcpro.2022.100217. |
| [13] | READ K A, POWELL M D, SREEKUMAR B K, et al. In vitro differentiation of effector CD4+ T helper cell subsets[J]. Methods Mol Biol, 2019, 1960:75-84. doi: 10.1007/978-1-4939-9167-9_6. |
| [14] | ZHU J. T helper cell differentiation,heterogeneity,and plasticity[J]. Cold Spring Harb Perspect Biol, 2018, 10(10):a030338. doi: 10.1101/cshperspect.a030338. |
| [15] | RUTERBUSCH M, PRUNER K B, SHEHATA L, et al. In vivo CD4+ T cell differentiation and function:Revisiting the Th1/Th2 paradigm[J]. Annu Rev Immunol, 2020, 38:705-725. doi: 10.1146/ annurev-immunol-103019-085803. |
| [16] | DZHALILOVA D S, KOSYREVA A M, DIATROPTOV M E, et al. Morphological characteristics of the thymus and spleen and the subpopulation composition of lymphocytes in peripheral blood during systemic inflammatory response in male rats with different resistance to hypoxia[J]. Int J Inflam, 2019, 2019:7584685. doi: 10.1155/2019/7584685. |
| [17] | CHEN Y, GABER T. Hypoxia/HIF modulates immune responses[J]. Biomedicines, 2021, 9(3):260. doi: 10.3390/biomedicines9030260. |
| [18] | LEE H S, JEONG G S. Salinosporamide A,a marine-derived proteasome inhibitor,inhibits T cell activation through regulating proliferation and the cell cycle[J]. Molecules, 2020, 25(21):5031. doi: 10.3390/molecules25215031. |
| [19] | HUBBI M E, SEMENZA G L. Regulation of cell proliferation by hypoxia-inducible factors[J]. Am J Physiol Cell Physiol, 2015, 309(12):C775-C782. doi: 10.1152/ajpcell.00279.2015. |
| [20] | CHO S H, RAYBUCK A L, STENGEL K, et al. Germinal centre hypoxia and regulation of antibody qualities by a hypoxia response system[J]. Nature, 2016, 537(7619):234-238. doi: 10.1038/nature19334. |
| [21] | SHAN T, CHEN S, CHEN X, et al. M2-TAM subsets altered by lactic acid promote T cell apoptosis through the PDL1/PD1 pathway[J]. Oncol Rep, 2020, 44(5):1885-1894. doi: 10.3892/or.2020.7767. |
| [22] | DHURIYA Y K, SHARMA D, NAIK A A. Cellular demolition:Proteins as molecular players of programmed cell death[J]. Int J Biol Macromol, 2019, 138:492-503. doi: 10.1016/j.ijbiomac.2019.07.113. |
| [23] | JENG P S, INOUE-YAMAUCHI A, HSIEH J J, et al. BH3-dependent and independent activation of BAX and BAK in mitochondrial apoptosis[J]. Curr Opin Physiol, 2018, 3:71-81. doi: 10.1016/j.cophys.2018.03.005. |
| [24] | YAMAZAKI T, GALLUZZI L. Bax and bak dynamics control mitochondrial DNA release during apoptosis[J]. Cell Death Differ, 2022, 29(6):1296-1298. doi: 10.1038/s41418-022-00985-2. |
| [25] | MUSAOGULLARI A, MANDATO A, CHAI Y C. Role of glutathione depletion and reactive oxygen species generation on caspase-3 activation:A study with the kinase inhibitor staurosporine[J]. Front Physiol, 2020, 11:998. doi: 10.3389/fphys.2020.00998. |
| [26] | WANG Y, LIU Y, FEI A, et al. Lncrna xist facilitates hypoxia-induced myocardial cell injury through targeting miR-191-5p/TRAF3 axis[J]. Mol Cell Biochem, 2022, 477(6):1697-1707. doi: 10.1007/s11010-022-04385-5. |
| [27] | ZHAO X, LIU L, LI R, et al. Hypoxia-inducible factor 1-α(HIF-1α)induces apoptosis of human uterosacral ligament fibroblasts through the death receptor and mitochondrial pathways[J]. Med Sci Monit, 2018, 24:8722-8733. doi: 10.12659/MSM.913384. |
| [1] | 杨晓芳, 贾新燕, 丰文君. miR-181a-5p通过HMGB1/NF-κB信号通路调控狼疮性肾炎小鼠肾小球系膜细胞增殖和凋亡[J]. 天津医药, 2026, 54(3): 232-237. |
| [2] | 张婧, 魏玉英, 宁海虹, 韦红梅, 王嘉玮, 曹薇, 吴宾. DUSP9在2型糖尿病心肌病小鼠心肌损伤中的保护作用及其机制[J]. 天津医药, 2026, 54(3): 238-244. |
| [3] | 王喆, 邱林, 马贲. 番茄来源胞外囊泡样颗粒对口腔鳞状细胞癌的作用效果研究[J]. 天津医药, 2026, 54(2): 145-150. |
| [4] | 李志伟, 张会超, 杨凤鸣, 曾垂义. 基于miR-144-3p/MAPK1通路探讨红参总皂苷对扩张型心肌病小鼠心肌细胞凋亡的影响[J]. 天津医药, 2026, 54(1): 23-29. |
| [5] | 赵兰君, 李良惠, 马馨, 巩娇娇, 郑臣辉, 石琳. 穿心莲内酯调控STAT3/GPX4通路对骨髓瘤细胞增殖和凋亡的影响[J]. 天津医药, 2026, 54(1): 8-13. |
| [6] | 孔翠文, 路延双, 孙丽萍, 于芬芬. LncRNA SNHG14靶向miR-30a-5p对高糖诱导的足细胞损伤的影响[J]. 天津医药, 2025, 53(9): 903-909. |
| [7] | 刘虹, 张玥玥, 王一琳, 王彩丽, 王晓敏, 毛敏, 李燕. MicroRNA-34a通过调控Wnt途径影响慢性淋巴细胞白血病进展的机制探讨[J]. 天津医药, 2025, 53(8): 785-790. |
| [8] | 万艳波, 刘明, 王勇. 秦皮甲素调节HMGB1/RAGE信号通路对缺氧/复氧诱导的心肌细胞损伤的影响[J]. 天津医药, 2025, 53(8): 796-801. |
| [9] | 刘海威, 杨洁, 王力, 蒙诗波, 唐旭松, 刘成仁, 王永旺. 木犀草素通过NFE2L2/x-CT/GPX4信号轴调控ROS水平抑制胶质母细胞瘤[J]. 天津医药, 2025, 53(7): 673-678. |
| [10] | 韩建存, 周谊. 川陈皮素调节FAK/AKT信号通路对喉鳞状细胞癌细胞增殖和凋亡的影响[J]. 天津医药, 2025, 53(6): 561-565. |
| [11] | 余朝霞, 马贲, 邱林, 高倩, 尼娜. 基于网络药理学和实验验证探究鲍式层孔菌多酚的抗头颈鳞癌机制[J]. 天津医药, 2025, 53(5): 456-461. |
| [12] | 李晨, 李占恩, 苏宏伟, 侯彩云, 董少文. KRT17调节Wnt/β-catenin信号通路对膀胱癌细胞增殖、凋亡及上皮间质转化的影响[J]. 天津医药, 2025, 53(5): 462-467. |
| [13] | 苏红见, 张春艳, 张卫东, 韩利, 乔亚红. 鸢尾素调控EBF3/ALOX15通路影响肺腺癌细胞增殖和迁移[J]. 天津医药, 2025, 53(4): 337-342. |
| [14] | 祁卫华, 黄广磊, 张媛媛, 班宏英, 毛诏旭. 连翘脂素调节cAMP/EPAC1/RAP1信号通路对肺癌细胞恶性进展的影响[J]. 天津医药, 2025, 53(4): 343-348. |
| [15] | 闫玲新, 李森, 郭改莉, 孟婉秋, 徐超. 异牡荆素通过miR-339-5p/HSPA8轴调节胰腺癌细胞的生物学行为[J]. 天津医药, 2025, 53(3): 230-235. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||