
天津医药 ›› 2024, Vol. 52 ›› Issue (1): 73-79.doi: 10.11958/20230179
收稿日期:2023-08-04
出版日期:2024-01-15
发布日期:2024-01-18
作者简介:杨阳(1988),女,讲师,主要从事生理学方面研究。E-mail:基金资助:Received:2023-08-04
Published:2024-01-15
Online:2024-01-18
杨阳, 何巧玉. 丹酚酸B对创伤后应激障碍模型大鼠认知功能和GSK-3β/β-Catenin信号通路的影响[J]. 天津医药, 2024, 52(1): 73-79.
YANG Yang, HE Qiaoyu. Impact of salvianolic acid B on cognitive function and GSK-3β/β-Catenin signaling pathway in rats with post-traumatic stress disorder[J]. Tianjin Medical Journal, 2024, 52(1): 73-79.
摘要:
目的 探讨丹酚酸B(Sal B)是否可通过调节糖原合成酶激酶-3β/β-连环蛋白(GSK-3β/β-Catenin)信号通路改善创伤后应激障碍(PTSD)模型大鼠认知功能。方法 60只大鼠随机分为正常组、PTSD组、Sal B低剂量组(10 mg/kg)、Sal B高剂量组(20 mg/kg)和GSK-3β抑制剂组(30 mg/kg CHIR-99021),每组12只。除正常组外,其余组大鼠采用单一延长应激法构建PTSD大鼠模型。旷场实验、Morris水迷宫实验评估大鼠认知功能;Nissl染色观察海马神经元病理学变化;TUNEL染色检测海马神经元凋亡;Western blot检测海马组织中裂解的胱天蛋白酶3(cleaved caspase-3)、B细胞淋巴瘤基因-2相关X蛋白(Bax)、原癌基因(c-Myc)、细胞周期蛋白D1(Cyclin D1)、总GSK-3β(t-GSK-3β)、磷酸化GSK-3β(p-GSK-3β)、总β-Catenin(t-β-Catenin)、磷酸化β-Catenin(p-β-Catenin)蛋白表达。结果 与PTSD组比较,Sal B低剂量组、Sal B高剂量组和GSK-3β抑制剂组大鼠爬行格数、站立次数、运动总距离、跨越原平台次数增多,逃避潜伏期、首次跨越原平台时间缩短,海马神经元凋亡率以及海马组织中Bax、cleaved caspase-3、t-GSK-3β、p-β-Catenin蛋白表达水平降低,Cyclin D1、c-Myc、p-GSK-3β、t-β-Catenin蛋白表达水平升高(P<0.05)。结论 Sal B能够减轻PTSD模型大鼠海马神经元凋亡、损伤并可改善其认知功能障碍,抑制GSK-3β/β-Catenin信号通路。
中图分类号:
| 组别 | 爬行格数/个 | 站立次数/次 | 运动总距离/cm |
|---|---|---|---|
| 正常组 | 76.33±4.68 | 14.25±1.46 | 2 436.18±90.29 |
| PTSD组 | 48.21±3.95a | 5.17±0.85a | 1 059.67±52.43a |
| Sal B低剂量组 | 61.83±4.40b | 9.08±1.02b | 1 836.29±71.85b |
| Sal B高剂量组 | 72.50±4.06bc | 12.47±1.25bc | 2 345.35±83.86bc |
| GSK-3β抑制剂组 | 69.58±4.12bc | 11.67±1.18bc | 2 308.42±89.27bc |
| F | 82.233** | 108.915** | 635.255** |
表1 各组大鼠旷场实验中爬行格数、站立次数和运动总距离比较
Tab.1 Comparison of the number of crawling cells, standing times and total distance of exercise in open field experiment between the five groups of rats (n=12,$\bar{x}±s$)
| 组别 | 爬行格数/个 | 站立次数/次 | 运动总距离/cm |
|---|---|---|---|
| 正常组 | 76.33±4.68 | 14.25±1.46 | 2 436.18±90.29 |
| PTSD组 | 48.21±3.95a | 5.17±0.85a | 1 059.67±52.43a |
| Sal B低剂量组 | 61.83±4.40b | 9.08±1.02b | 1 836.29±71.85b |
| Sal B高剂量组 | 72.50±4.06bc | 12.47±1.25bc | 2 345.35±83.86bc |
| GSK-3β抑制剂组 | 69.58±4.12bc | 11.67±1.18bc | 2 308.42±89.27bc |
| F | 82.233** | 108.915** | 635.255** |
| 组别 | 1 d | 2 d |
|---|---|---|
| 正常组 | 31.59±4.42 | 23.47±4.03 |
| PTSD组 | 75.64±6.83a | 70.95±6.92a |
| Sal B低剂量组 | 56.78±5.97b | 49.26±5.84b |
| Sal B高剂量组 | 43.15±5.62bc | 35.24±5.16bc |
| GSK-3β抑制剂组 | 46.06±4.98bc | 37.58±5.34bc |
| F | 104.545** | 126.256** |
| 组别 | 3 d | 4 d |
| 正常组 | 15.26±3.15 | 8.95±2.64 |
| PTSD组 | 67.08±6.54a | 61.24±6.71a |
| Sal B低剂量组 | 41.65±5.23b | 33.17±4.38b |
| Sal B高剂量组 | 30.57±4.31bc | 24.97±3.97bc |
| GSK-3β抑制剂组 | 33.19±4.58bc | 27.12±3.65bc |
| F | 182.284** | 217.942** |
表2 各组大鼠Morris水迷宫实验中逃避潜伏期比较
Tab.2 Comparison of escape latency in Morris water maze experiment between the five groups of rats (n=12,s,$\bar{x}±s$)
| 组别 | 1 d | 2 d |
|---|---|---|
| 正常组 | 31.59±4.42 | 23.47±4.03 |
| PTSD组 | 75.64±6.83a | 70.95±6.92a |
| Sal B低剂量组 | 56.78±5.97b | 49.26±5.84b |
| Sal B高剂量组 | 43.15±5.62bc | 35.24±5.16bc |
| GSK-3β抑制剂组 | 46.06±4.98bc | 37.58±5.34bc |
| F | 104.545** | 126.256** |
| 组别 | 3 d | 4 d |
| 正常组 | 15.26±3.15 | 8.95±2.64 |
| PTSD组 | 67.08±6.54a | 61.24±6.71a |
| Sal B低剂量组 | 41.65±5.23b | 33.17±4.38b |
| Sal B高剂量组 | 30.57±4.31bc | 24.97±3.97bc |
| GSK-3β抑制剂组 | 33.19±4.58bc | 27.12±3.65bc |
| F | 182.284** | 217.942** |
| 组别 | 跨越原平台 次数/次 | 首次跨越原 平台时间/s |
|---|---|---|
| 正常组 | 9.25±1.38 | 10.68±1.29 |
| PTSD组 | 3.42±0.85a | 39.45±3.18a |
| Sal B低剂量组 | 4.83±0.96b | 27.84±2.86b |
| Sal B高剂量组 | 7.67±1.25bc | 19.07±1.95bc |
| GSK-3β抑制剂组 | 7.00±1.04bc | 21.31±2.42bc |
| F | 51.921** | 233.936** |
表3 各组大鼠Morris水迷宫实验中跨越原平台次数和首次跨越原平台时间比较
Tab.3 Comparison of the times of crossing the original platform and the time of crossing the original platform for the first time in Morris water maze experiment between the five groups of rats (n=12,$\bar{x}±s$)
| 组别 | 跨越原平台 次数/次 | 首次跨越原 平台时间/s |
|---|---|---|
| 正常组 | 9.25±1.38 | 10.68±1.29 |
| PTSD组 | 3.42±0.85a | 39.45±3.18a |
| Sal B低剂量组 | 4.83±0.96b | 27.84±2.86b |
| Sal B高剂量组 | 7.67±1.25bc | 19.07±1.95bc |
| GSK-3β抑制剂组 | 7.00±1.04bc | 21.31±2.42bc |
| F | 51.921** | 233.936** |
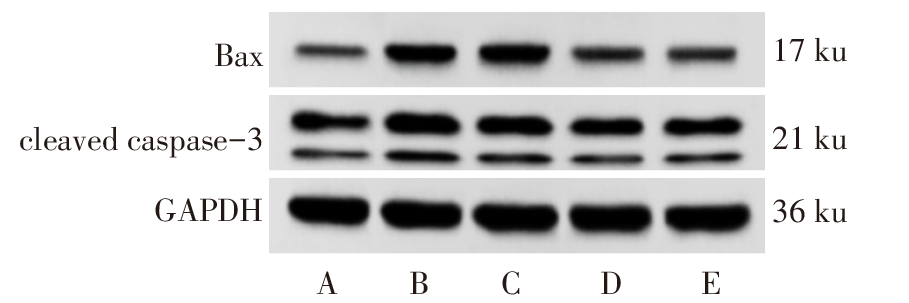
图3 Western blot检测各组大鼠海马组织中Bax、cleaved caspase-3蛋白表达 A:正常组;B:PTSD组;C:Sal B低剂量组;D:Sal B高剂量组;E:GSK-3β抑制剂组。
Fig.3 The expression of Bax and cleaved caspase-3 protein in hippocampus of each group detected by Western blot assay
| 组别 | 神经元凋亡率/ % | Bax | cleaved caspase-3 |
|---|---|---|---|
| 正常组 | 5.89±0.96 | 0.18±0.04 | 0.27±0.05 |
| PTSD组 | 57.26±7.85a | 0.79±0.06a | 0.92±0.08a |
| Sal B低剂量组 | 32.17±5.43b | 0.51±0.06b | 0.63±0.06b |
| Sal B高剂量组 | 20.42±2.56bc | 0.29±0.05bc | 0.41±0.05bc |
| GSK-3β抑制剂组 | 22.05±3.18bc | 0.32±0.06bc | 0.45±0.07bc |
| F | 100.345** | 115.510** | 94.342** |
表4 各组大鼠海马神经元凋亡率以及海马组织中Bax、cleaved caspase-3蛋白表达水平比较
Tab.4 Comparison of apoptosis rate of hippocampal neurons and expression levels of Bax and cleaved caspase-3 in hippocampal tissue between the five groups of rats (n=6,$\bar{x}±s$)
| 组别 | 神经元凋亡率/ % | Bax | cleaved caspase-3 |
|---|---|---|---|
| 正常组 | 5.89±0.96 | 0.18±0.04 | 0.27±0.05 |
| PTSD组 | 57.26±7.85a | 0.79±0.06a | 0.92±0.08a |
| Sal B低剂量组 | 32.17±5.43b | 0.51±0.06b | 0.63±0.06b |
| Sal B高剂量组 | 20.42±2.56bc | 0.29±0.05bc | 0.41±0.05bc |
| GSK-3β抑制剂组 | 22.05±3.18bc | 0.32±0.06bc | 0.45±0.07bc |
| F | 100.345** | 115.510** | 94.342** |
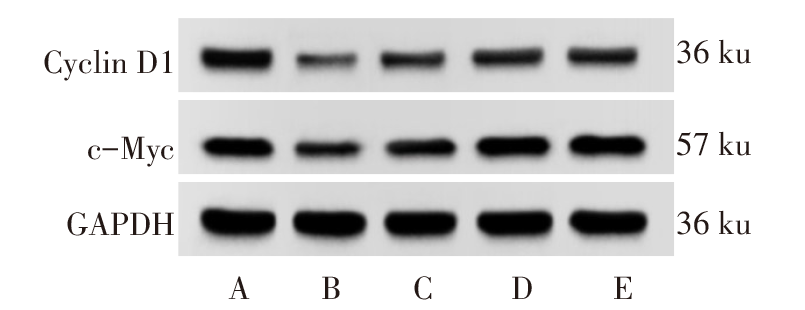
图4 Western blot检测各组大鼠海马组织中Cyclin D1、c-Myc蛋白表达 A:正常组;B:PTSD组;C:Sal B低剂量组;D:Sal B高剂量组;E:GSK-3β抑制剂组。
Fig.4 The expression of Cyclin D1 and c-Myc protein in hippocampal tissue of rats in each group detected by Western blot assay
| 组别 | Cyclin D1 | c-Myc |
|---|---|---|
| 正常组 | 0.69±0.06 | 0.57±0.05 |
| PTSD组 | 0.23±0.04a | 0.18±0.03a |
| Sal B低剂量组 | 0.40±0.05b | 0.35±0.03b |
| Sal B高剂量组 | 0.54±0.05bc | 0.46±0.04bc |
| GSK-3β抑制剂组 | 0.51±0.06bc | 0.42±0.05bc |
| F | 63.761** | 74.750** |
表5 各组大鼠海马组织中c-Myc、Cyclin D1蛋白表达水平比较
Tab.5 Comparison of protein expression levels of c-Myc and Cyclin D1 in hippocampal tissue between the five groups of rats (n=6,$\bar{x}±s$)
| 组别 | Cyclin D1 | c-Myc |
|---|---|---|
| 正常组 | 0.69±0.06 | 0.57±0.05 |
| PTSD组 | 0.23±0.04a | 0.18±0.03a |
| Sal B低剂量组 | 0.40±0.05b | 0.35±0.03b |
| Sal B高剂量组 | 0.54±0.05bc | 0.46±0.04bc |
| GSK-3β抑制剂组 | 0.51±0.06bc | 0.42±0.05bc |
| F | 63.761** | 74.750** |
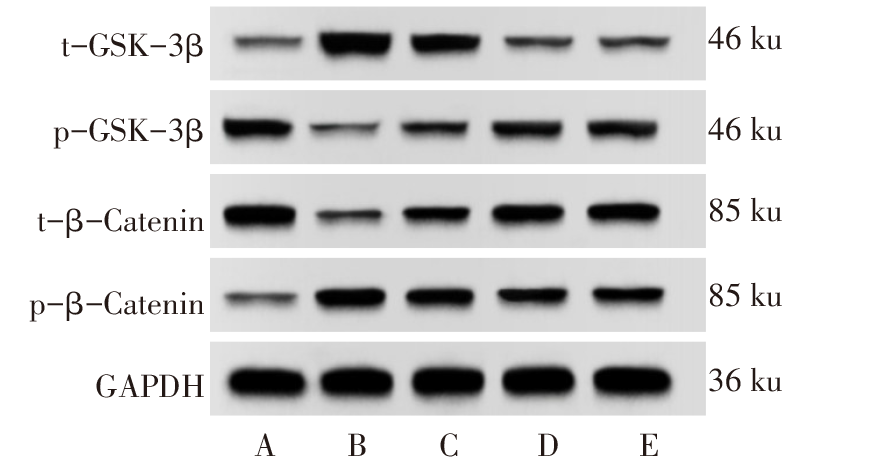
图5 Western blot检测各组大鼠海马组织中t-GSK-3β、p-GSK-3β、t-β-Catenin、p-β-Catenin蛋白表达情况 A:正常组;B:PTSD组;C:Sal B低剂量组;D:Sal B高剂量组;E:GSK-3β抑制剂组。
Fig.5 The expression of t-GSK-3β, p-GSK-3β, t-β-Catenin and p-β-Catenin in hippocampal tissue of rats in each group detected by Western blot assay
| 组别 | t-GSK- 3β | p-GSK- 3β | t-β- Catenin | p-β- Catenin |
|---|---|---|---|---|
| 正常组 | 0.15±0.03 | 0.61±0.05 | 0.79±0.05 | 0.18±0.03 |
| PTSD组 | 0.78±0.06a | 0.18±0.03a | 0.23±0.04a | 0.67±0.06a |
| Sal B低剂量组 | 0.54±0.05b | 0.35±0.04b | 0.51±0.05b | 0.49±0.05b |
| Sal B高剂量组 | 0.37±0.04bc | 0.45±0.04bc | 0.65±0.05bc | 0.35±0.04bc |
| GSK-3β抑制 剂组 | 0.34±0.04bc | 0.47±0.05bc | 0.66±0.06bc | 0.33±0.05bc |
| F | 165.088** | 83.802** | 107.528** | 92.378** |
表6 各组大鼠海马组织中t-GSK-3β、p-GSK-3β、t-β-Catenin、p-β-Catenin蛋白表达水平比较
Tab.6 Comparison of protein expression levels of t-GSK-3β, p-GSK-3β, t-β-Catenin and p-β-Catenin in hippocampal tissue between the five groups of rats (n=6,$\bar{x}±s$)
| 组别 | t-GSK- 3β | p-GSK- 3β | t-β- Catenin | p-β- Catenin |
|---|---|---|---|---|
| 正常组 | 0.15±0.03 | 0.61±0.05 | 0.79±0.05 | 0.18±0.03 |
| PTSD组 | 0.78±0.06a | 0.18±0.03a | 0.23±0.04a | 0.67±0.06a |
| Sal B低剂量组 | 0.54±0.05b | 0.35±0.04b | 0.51±0.05b | 0.49±0.05b |
| Sal B高剂量组 | 0.37±0.04bc | 0.45±0.04bc | 0.65±0.05bc | 0.35±0.04bc |
| GSK-3β抑制 剂组 | 0.34±0.04bc | 0.47±0.05bc | 0.66±0.06bc | 0.33±0.05bc |
| F | 165.088** | 83.802** | 107.528** | 92.378** |
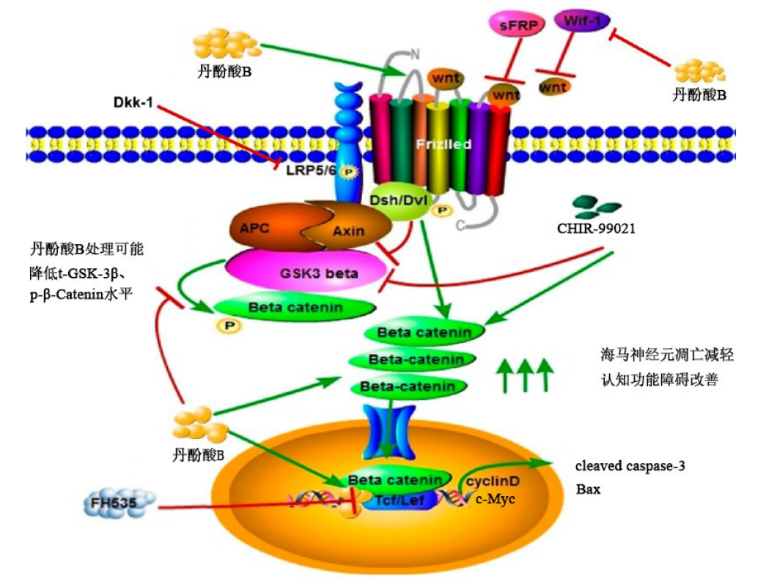
图6 Sal B的信号通路图 Tcf/Lef:T细胞因子/淋巴细胞增强因子;FH535:Tcf抑制剂;Dkk-1:Wnt抑制剂;sFRP:分泌性卷曲相关蛋白;Wif-1:Wnt抑制因子1;APC:Wnt信号通路调节因子;Axin:轴蛋白;Dsh/Dvl:蓬乱蛋白/蓬乱蛋白Dsh同源物2;Frizzled:卷曲蛋白;LRP-5/6:低密度脂蛋白受体相关蛋白5/6。
Fig.6 Signal path diagram of Sal B
| [1] | FENSTER R J, LEBOIS L, RESSLER K J, et al. Brain circuit dysfunction in post-traumatic stress disorder: from mouse to man[J]. Nat Rev Neurosci, 2018, 19(9):535-551. doi:10.1038/s41583-018-0039-7. |
| [2] | MAERCKER A, CLOITRE M, BACHEM R, et al. Complex post-traumatic stress disorder[J]. Lancet, 2022, 400(10345):60-72. doi:10.1016/S0140-6736(22)00821-2. |
| [3] | MERIANS A N, SPILLER T, HARPAZ-ROTEM I, et al. Post-traumatic stress disorder[J]. Med Clin North Am, 2023, 107(1):85-99. doi:10.1016/j.mcna.2022.04.003. |
| [4] | YANG Y, SONG J, LIU N, et al. Salvianolic acid A relieves cognitive disorder after chronic cerebral ischemia: Involvement of Drd2/Cryab/NF-κB pathway[J]. Pharmacol Res, 2022, 175:105989. doi:10.1016/j.phrs.2021.105989. |
| [5] | 李建, 李强, 李延峰, 等. 丹酚酸B治疗阿尔茨海默病的作用机制研究进展[J]. 中国药理学通报, 2022, 38(4):487-491. |
| LI J, LI Q, LI Y F, et al. Recent progress of salvianolic acid B for treatment of Alzheimer’s disease[J]. Chinese Pharmacological Bulletin, 2022, 38(4):487-491. doi:10.12360/CPB202108020. | |
| [6] | HUI J, ZHANG J, PU M, et al. Modulation of GSK-3β/β-catenin signaling contributes to learning and memory impairment in a rat model of depression[J]. Int J Neuropsychopharmacol, 2018, 21(9):858-870. doi:10.1093/ijnp/pyy040. |
| [7] | ZHANG S, KONG D W, MA G D, et al. Long-term administration of salvianolic acid A promotes endogenous neurogenesis in ischemic stroke rats through activating Wnt3a/GSK3β/β-catenin signaling pathway[J]. Acta Pharmacol Sin, 2022, 43(9):2212-2225. doi:10.1038/s41401-021-00844-9. |
| [8] | SHU T, LIU C, PANG M, et al. Salvianolic acid B promotes neural differentiation of induced pluripotent stem cells via PI3K/AKT/GSK3β/β-catenin pathway[J]. Neurosci Lett, 2018, 671:154-160. doi:10.1016/j.neulet.2018.02.007. |
| [9] | 丛海涛, 丁进峰, 何海娟, 等. 右美托咪定对创伤后应激障碍大鼠核因子κB抑制蛋白激酶/核因子κB抑制蛋白α/核因子κB通路及认知功能障碍的影响[J]. 解剖学报, 2022, 53(3):295-301. |
| CONG H T, DING J F, HE H J, et al. Effects of dexmedetomidine on nuclear factor-κB inhibitor protein kinase/nuclear factor-κB inhibitor proteinα/nuclear factor-κB pathway and cognitive dysfunction in rats with post-traumatic stress disorder[J]. Acta Anatomica Sinica, 2022, 53(3):295-301. doi:10.16098/j.issn.0529-1356.2022.03.004. | |
| [10] | ALQURAAN L, ALZOUBI K H, HAMMAD H, et al. Omega-3 fatty acids prevent post-traumatic stress disorder-induced memory impairment[J]. Biomolecules, 2019, 9(3):100. doi:10.3390/biom9030100. |
| [11] | 孙一萍, 李晓艳, 邵瑞洁, 等. 针刺对创伤后应激障碍大鼠海马内质网应激相关分子的影响[J]. 针刺研究, 2022, 47(3):224-230. |
| SUN Y P, LI X Y, SHAO R J, et al. Effect of acupuncture on endoplasmic reticulum stress-related factors in hippocampus of post-traumatic stress disorder rats[J]. Acupuncture Research, 2022, 47(3):224-230. doi:10.13702/j.1000-0607.20210718. | |
| [12] | ZHANG X, WU Q, LU Y, et al. Cerebroprotection by salvianolic acid B after experimental subarachnoid hemorrhage occurs via Nrf2- and SIRT1-dependent pathways[J]. Free Radic Biol Med, 2018, 124:504-516. doi:10.1016/j.freeradbiomed.2018.06.035. |
| [13] | KONDASHEVSKAYA M V, ARTEM'YEVA K A, ALEKSANKINA V V, et al. Phenotypically determined liver dysfunction in a Wistar rat model of post-traumatic stress disorder[J]. J Evol Biochem Physiol, 2022, 58(4):1015-1024. doi:10.1134/S002209302204007X. |
| [14] | SUR B, LEE B. Ginsenoside Rg3 modulates spatial memory and fear memory extinction by the HPA axis and BDNF-TrkB pathway in a rat post-traumatic stress disorder[J]. J Nat Med, 2022, 76(4):821-831. doi:10.1007/s11418-022-01636-z. |
| [15] | JIA Y, HAN Y, WANG X, et al. Role of apoptosis in the post-traumatic stress disorder model-single prolonged stressed rats[J]. Psychoneuroendocrinology, 2018, 95:97-105. doi:10.1016/j.psyneuen.2018.05.015. |
| [16] | SEO J H, PARK H S, PARK S S, et al. Physical exercise ameliorates psychiatric disorders and cognitive dysfunctions by hippocampal mitochondrial function and neuroplasticity in post-traumatic stress disorder[J]. Exp Neurol, 2019, 322:113043. doi:10.1016/j.expneurol.2019.113043. |
| [17] | XIAO Z, LIU W, MU Y P, et al. Pharmacological effects of salvianolic acid B against oxidative damage[J]. Front Pharmacol, 2020, 11:572373. doi:10.3389/fphar.2020.572373. |
| [18] | GUO S S, WANG Z G. Salvianolic acid B from Salvia miltiorrhiza bunge:a potential antitumor agent[J]. Front Pharmacol, 2022, 13:1042745. doi:10.3389/fphar.2022.1042745. |
| [19] | ZHAO R, LIU X, ZHANG L, et al. Current progress of research on neurodegenerative diseases of salvianolic acid B[J]. Oxid Med Cell Longev, 2019, 2019:3281260. doi:10.1155/2019/3281260. |
| [20] | YU X, GUAN Q, WANG Y, et al. Anticonvulsant and anti-apoptosis effects of salvianolic acid B on pentylenetetrazole-kindled rats via AKT/CREB/BDNF signaling[J]. Epilepsy Res, 2019, 154:90-96. doi:10.1016/j.eplepsyres.2019.05.007. |
| [21] | ZHAO Y, ZHANG Y, ZHANG J, et al. Salvianolic acid B protects against MPP+-induced neuronal injury via repressing oxidative stress and restoring mitochondrial function[J]. Neuroreport, 2021, 32(9):815-823. doi:10.1097/WNR.0000000000001660. |
| [22] | YANG Y, WANG L, ZHANG C, et al. Ginsenoside Rg1 improves Alzheimer's disease by regulating oxidative stress, apoptosis, and neuroinflammation through Wnt/GSK-3β/β-catenin signaling pathway[J]. Chem Biol Drug Des, 2022, 99(6):884-896. doi:10.1111/cbdd.14041. |
| [23] | HUANG Y L, ZHANG J N, HOU T Z, et al. Inhibition of Wnt/β-catenin signaling attenuates axonal degeneration in models of Parkinson's disease[J]. Neurochem Int, 2022, 159:105389. doi:10.1016/j.neuint.2022.105389. |
| [24] | MAI C L, WEI X, GUI W S, et al. Differential regulation of GSK-3β in spinal dorsal horn and in hippocampus mediated by interleukin-1beta contributes to pain hypersensitivity and memory deficits following peripheral nerve injury[J]. Mol Pain, 2019, 15:1744806919826789. doi:10.1177/1744806919826789. |
| [25] | WANG Y, AN X, ZHANG X, et al. Lithium chloride ameliorates cognition dysfunction induced by sevoflurane anesthesia in rats[J]. FEBS Open Bio, 2020, 10(2):251-258. doi:10.1002/2211-5463.12779. |
| [26] | 钱红月, 肖移生, 侯吉华, 等. 黄精丸对D-半乳糖和冈田酸所致学习记忆障碍小鼠海马Wnt/β-catenin信号通路相关蛋白表达的影响[J]. 中国实验方剂学杂志, 2021, 27(1):63-71. |
| QIAN H Y, XIAO Y S, HOU J H, et al. Effect of Huangjingwan on expressions of Wnt/β-catenin signal pathway-associated proteins in hippocampus of mice with Alzheimer's Disease induced by D-galactose and okadaic acid with learning and memory disorders[J]. Chinese Journal of Experimental Traditional Medical Formulae, 2021, 27(1):63-71. doi:10.13422/j.cnki.syfjx.20201876. | |
| [27] | ZHOU H, LIU Y, SUN L, et al. Salvianolic acid B activates Wnt/β-catenin signaling following spinal cord injury[J]. Exp Ther Med, 2020, 19(2):825-832. doi:10.3892/etm.2019.8292. |
| [28] | WANG B, KHAN S, WANG P, et al. A Highly Selective GSK-3β Inhibitor CHIR99021 promotes osteogenesis by activating canonical and autophagy-mediated Wnt signaling[J]. Front Endocrinol(Lausanne), 2022, 13:926622. doi:10.3389/fendo.2022.926622. |
| [29] | KHURANA C, BEDI O. Proposed hypothesis of GSK-3β inhibition for stimulating Wnt/β-catenin signaling pathway which triggers liver regeneration process[J]. Naunyn Schmiedebergs Arch Pharmacol, 2022, 395(3):377-380. doi:10.1007/s00210-022-02207-5. |
| [1] | 杨晓芳, 贾新燕, 丰文君. miR-181a-5p通过HMGB1/NF-κB信号通路调控狼疮性肾炎小鼠肾小球系膜细胞增殖和凋亡[J]. 天津医药, 2026, 54(3): 232-237. |
| [2] | 张婧, 魏玉英, 宁海虹, 韦红梅, 王嘉玮, 曹薇, 吴宾. DUSP9在2型糖尿病心肌病小鼠心肌损伤中的保护作用及其机制[J]. 天津医药, 2026, 54(3): 238-244. |
| [3] | 王喆, 邱林, 马贲. 番茄来源胞外囊泡样颗粒对口腔鳞状细胞癌的作用效果研究[J]. 天津医药, 2026, 54(2): 145-150. |
| [4] | 李志伟, 张会超, 杨凤鸣, 曾垂义. 基于miR-144-3p/MAPK1通路探讨红参总皂苷对扩张型心肌病小鼠心肌细胞凋亡的影响[J]. 天津医药, 2026, 54(1): 23-29. |
| [5] | 赵兰君, 李良惠, 马馨, 巩娇娇, 郑臣辉, 石琳. 穿心莲内酯调控STAT3/GPX4通路对骨髓瘤细胞增殖和凋亡的影响[J]. 天津医药, 2026, 54(1): 8-13. |
| [6] | 孔翠文, 路延双, 孙丽萍, 于芬芬. LncRNA SNHG14靶向miR-30a-5p对高糖诱导的足细胞损伤的影响[J]. 天津医药, 2025, 53(9): 903-909. |
| [7] | 周玉科, 孙伟, 高庆, 贺军. 七氟醚调节ROS/TXNIP/NLRP3通路对脊髓损伤大鼠神经细胞炎症的影响[J]. 天津医药, 2025, 53(9): 910-915. |
| [8] | 万艳波, 刘明, 王勇. 秦皮甲素调节HMGB1/RAGE信号通路对缺氧/复氧诱导的心肌细胞损伤的影响[J]. 天津医药, 2025, 53(8): 796-801. |
| [9] | 刘海威, 杨洁, 王力, 蒙诗波, 唐旭松, 刘成仁, 王永旺. 木犀草素通过NFE2L2/x-CT/GPX4信号轴调控ROS水平抑制胶质母细胞瘤[J]. 天津医药, 2025, 53(7): 673-678. |
| [10] | 陈国庆, 严琴, 陈明九, 王允琴. 撮风散加味治疗前循环脑梗死急性期风痰阻络证的临床研究[J]. 天津医药, 2025, 53(7): 761-764. |
| [11] | 韩建存, 周谊. 川陈皮素调节FAK/AKT信号通路对喉鳞状细胞癌细胞增殖和凋亡的影响[J]. 天津医药, 2025, 53(6): 561-565. |
| [12] | 祁卫华, 黄广磊, 张媛媛, 班宏英, 毛诏旭. 连翘脂素调节cAMP/EPAC1/RAP1信号通路对肺癌细胞恶性进展的影响[J]. 天津医药, 2025, 53(4): 343-348. |
| [13] | 贾方, 李平, 李静, 尹路. 全膝关节置换术后神经认知功能障碍的影响因素及列线图模型的构建[J]. 天津医药, 2025, 53(3): 287-292. |
| [14] | 马莉莉, 李子沐, 王亮, 许彭, 李秀梅. 间充质干细胞外泌体对食管癌ECA109细胞生物学行为的影响[J]. 天津医药, 2025, 53(2): 113-117. |
| [15] | 吴宾, 杨自更, 张婧, 李书红, 余凤, 王嘉玮, 李彩玲. 柚皮素对低氧性肺动脉高压大鼠右心室重塑的影响[J]. 天津医药, 2025, 53(2): 129-134. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||
